ADME Toxicology Testing Market Size, Share & Trends Analysis Report By Technology (Cell Culture, High Throughput, Molecular Imaging, OMICS Technology), By Testing Method (Cellular Assay, Biochemical Assay, In-Silica, Ex-vivo), By Application (Systemic Toxicity, Renal Toxicity, Hepatotoxicity, Neurotoxicity, Other Toxicities) and By Region (North America, Europe, APAC, Middle East and Africa, LATAM) Forecasts, 2026-2034
What is the Size of ADME Toxicology Testing Market?
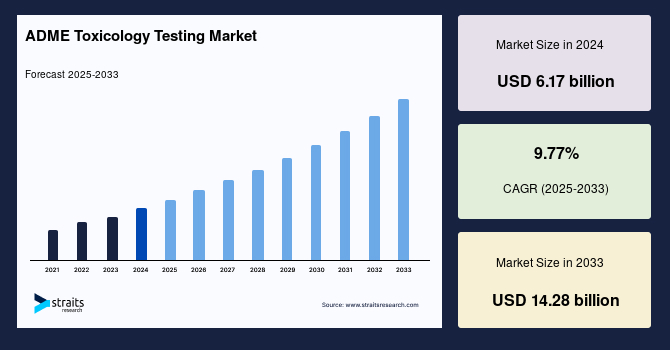
The ADME toxicology testing market size was valued at USD 7.00 billion in 2025 and is projected to grow from USD 7.69 billion in 2026 to USD 16.78 billion by 2034 at a CAGR of 10.24% during the forecast period (2026-2034).
Key Market Insights
- North America dominated the ADME toxicology testing market with the largest share of 40.02% in 2025.
- The Asia Pacific is expected to be the fastest-growing region in the ADME toxicology testing market during the forecast period at a CAGR of 12.24%.
- Based on technology, the cell culture segment dominated the market with a revenue share of 43.12% in 2025.
- Based on testing method, the cellular assay segment dominated the market with a revenue share of 48.23% in 2025.
- Based on application, the systematic toxicity segment dominated the market with a share of 50.21% in 2025.
- The US ADME toxicology testing market size was valued at USD 2.42 billion in 2025 and is projected to reach USD 2.66 billion in 2026.
Market Summary
| Market Metric | Details & Data (2025-2034) |
|---|---|
| 2025 Market Valuation | USD 7.00 billion |
| Estimated 2026 Value | USD 7.69 billion |
| Projected 2034 Value | USD 16.78 billion |
| CAGR (2026-2034) | 10.24% |
| Study Period | 2022-2034 |
| Dominant Region | North America |
| Fastest Growing Region | Asia Pacific |
| Key Market Players | Symeres, Promega Corporation, PerkinElmer Inc., Agilent Technologies, Corning Inc. |
Download Free Sample Report to Get Detailed Insights.
What are the Latest Trends in ADME Toxicology Testing Market?
The integration of microphysiological systems such as organ-on-chip platforms is emerging as a major market trend, as these models enable more physiologically relevant simulation of human drug metabolism and toxicity compared to conventional in vitro assays, thereby encouraging pharmaceutical companies to incorporate advanced predictive platforms in early drug discovery workflows. This trend compels testing service providers and technology developers to invest in sophisticated microfluidic technologies and human-cell-based models to improve predictive accuracy and remain competitive.
The growing use of artificial intelligence and machine-learning-driven predictive modeling in ADME toxicology analysis uses computational tools with experimental data to forecast absorption, metabolic pathways, and toxicity risks earlier in the development cycle. This shift enables pharmaceutical developers to reduce experimental iterations while encouraging contract research organizations to expand bioinformatics and computational toxicology capabilities.
Increasing outsourcing of ADME toxicology testing helps specialized contract research organizations as pharmaceutical and biotechnology companies seek to accelerate development timelines while managing internal infrastructure costs. This outsourcing trend is pushing service providers to expand integrated ADME-tox testing platforms that combine in vitro, in vivo, and in silico services within a single workflow.
What are the Key Drivers of ADME Toxicology Testing Market?
The growing complexity of modern drug candidates, particularly large molecules, antibody-drug conjugates, and targeted therapies, is a major market driver because these advanced therapeutics require detailed metabolic and toxicity profiling that traditional screening approaches cannot adequately address. This complexity encourages drug developers to adopt comprehensive ADME toxicology testing programs and stimulates demand for advanced analytical technologies and specialized testing expertise.
The rising emphasis on early-stage safety profiling in pharmaceutical research pipelines drives the market growth, as companies increasingly recognize that identifying pharmacokinetic limitations and toxicity liabilities in preclinical stages significantly reduces costly late-stage clinical failures. As a result, drug developers are investing more heavily in robust ADME toxicology assessments, which increases demand for testing platforms capable of providing early predictive safety insights.
The expansion of global pharmaceutical research pipelines, particularly among biotechnology startups and emerging drug developers, also drives the market because smaller companies often lack in-house toxicology infrastructure and depend heavily on external ADME testing services. This creates consistent demand for specialized laboratories capable of supporting early discovery and preclinical development programs.
Which Factors are Limiting the Growth of ADME Toxicology Testing Market?
The major restraint in the market is the limited translational predictability between preclinical models and human physiology, as in vitro systems and animal models do not always accurately replicate human metabolic pathways or toxicity responses. This uncertainty can lead pharmaceutical companies to repeat or supplement studies with additional models, increasing development costs and slowing decision-making for testing service providers.
The high cost associated with advanced ADME toxicology platforms, including high-resolution mass spectrometry, high-throughput screening systems, and specialized cell culture models, restrains the market growth. These capital-intensive technologies can restrict the ability of smaller laboratories and emerging research institutions to adopt advanced testing capabilities, limiting the overall expansion of testing infrastructure.
Regulatory variability across different global markets also restrains the industry, as drug developers must often tailor ADME toxicology studies to meet diverse regional regulatory expectations regarding safety data and testing methodologies. This fragmentation complicates study design and increases the operational burden for testing providers that support multinational drug development programs.
What are the Growth Opportunities for Players in ADME Toxicology Testing Market?
The increasing development of gene therapies, RNA-based medicines, and cell-based therapeutics presents a significant opportunity for the ADME toxicology testing market because these novel modalities require entirely new frameworks for evaluating biodistribution, metabolic stability, and long-term toxicity. This creates opportunities for testing providers to design specialized assays and analytical platforms tailored to emerging therapeutic classes.
A major opportunity lies in the adoption of human-derived 3D tissue cultures and induced pluripotent stem cell-based models for metabolic and toxicity evaluation, as these platforms can improve human relevance while reducing reliance on animal testing. Companies developing these advanced biological models can expand their role in drug safety testing by offering more predictive preclinical screening solutions.
The growing regulatory and ethical push toward reducing animal experimentation also opens avenues for innovative in vitro and computational ADME toxicology approaches, as regulatory bodies increasingly encourage alternative testing strategies. This transition allows technology developers and contract testing organizations to expand validated non-animal testing platforms that meet regulatory expectations while improving testing efficiency.
Regional Analysis
North America ADME Toxicology Testing Market
North America held a significant share of the ADME toxicology testing market in 2025, with a 40.02% share, due to its highly structured drug development regulatory framework and strong pharmaceutical innovation ecosystem. In the US, regulatory agencies such as the US Food and Drug Administration (FDA) require extensive pharmacology and toxicology data before a drug candidate can proceed to human clinical trials through the Investigational New Drug (IND) application process. These preclinical studies evaluate pharmacokinetics and toxicity to determine whether a compound is safe for initial testing in humans, which directly drives the demand for ADME and toxicology testing services across pharmaceutical pipelines.
Additionally, the region hosts a large concentration of global pharmaceutical companies, biotechnology startups, and specialized contract research organizations that conduct preclinical safety studies under Good Laboratory Practice (GLP) standards, further strengthening the regional demand for advanced ADME testing platforms.
Asia Pacific ADME Toxicology Testing Market
The Asia Pacific market is anticipated to register a CAGR of 12.24%, driven by increasing pharmaceutical R&D investments and strong government initiatives supporting biotechnology development. Several countries in the region, including China and India, have implemented policy programs and funding initiatives aimed at strengthening domestic pharmaceutical manufacturing and drug discovery capabilities. For example, India introduced a production-linked incentive program allocating billions of dollars to support pharmaceutical innovation and manufacturing expansion, which indirectly increases demand for preclinical ADME and toxicology evaluation services.
In addition, the number of early-phase clinical trials in China has increased significantly over the past decade due to government support and improved research infrastructure, which requires extensive preclinical safety testing prior to human trials. These factors collectively encourage pharmaceutical companies to conduct ADME and toxicity testing within the region.
Europe ADME Toxicology Testing Market
The market in Europe is growing due to the region’s strong regulatory oversight and emphasis on standardized laboratory research practices. European pharmaceutical research institutions and CROs operate under internationally recognized Good Laboratory Practice (GLP) frameworks that govern how non-clinical toxicology and safety studies are conducted and reported to regulatory authorities. These standards ensure the reliability of toxicity data used in drug approval applications, encouraging pharmaceutical developers to invest in rigorous ADME testing programs within European laboratories. The region has a strong academic-industry collaboration environment that supports translational drug research, enabling universities, biotechnology firms, and contract research organizations to collectively conduct preclinical metabolism and toxicity studies required before clinical evaluation.
Middle East & Africa ADME Toxicology Testing Market
The Middle East & Africa market is developing gradually as governments invest in biomedical research infrastructure and healthcare innovation programs. Countries such as the United Arab Emirates and Saudi Arabia have launched national research strategies aimed at expanding biotechnology capabilities and supporting pharmaceutical research collaborations with global companies. As these countries increase investments in life science research institutions and specialized laboratories, demand for preclinical safety testing services, including drug metabolism and toxicity analysis, is gradually emerging within regional research ecosystems. Moreover, growing clinical research activity and collaborations with international pharmaceutical companies are encouraging regional laboratories to establish capabilities in toxicology and pharmacokinetic testing.
Latin America ADME Toxicology Testing Market
The Latin America market is expanding due to the region’s increasing participation in global pharmaceutical research and clinical trial activities. Countries such as Brazil, Mexico, and Argentina are strengthening regulatory oversight of drug development while encouraging international pharmaceutical companies to conduct clinical studies in the region. As drug developers prepare compounds for these clinical trials, preclinical pharmacokinetic and toxicity assessments become necessary to meet regulatory and safety requirements before human testing begins. The presence of a growing network of research universities, public health institutes, and emerging contract research organizations is gradually improving the region’s capacity to conduct ADME and toxicology studies as part of multinational drug development programs.
Technology Insights
Cell culture dominated the technology segment with a share of 43.12% because it provides a controlled biological environment to evaluate drug absorption, metabolism, and cytotoxicity using human or animal-derived cells. Pharmaceutical and biotechnology companies widely rely on cell-based systems such as hepatocytes, intestinal epithelial cells, and renal cell lines to replicate organ-specific metabolic pathways and assess compound safety during early-stage drug discovery, which further strengthens the dominance of the cell culture technology segment.
The high-throughput technologies segment is expected to grow at a CAGR of 11.23%, driven by the increasing need to screen large chemical libraries efficiently during early drug discovery. Automated screening platforms integrated with robotics, miniaturized assays, and high-content detection systems enable researchers to evaluate thousands of compounds simultaneously for metabolic stability, permeability, and toxicity endpoints. As pharmaceutical companies aim to accelerate candidate selection and reduce development timelines, the demand for high-throughput ADME screening platforms continues to expand rapidly.
Testing Method Insights
Cellular assays dominated the testing method segment with a share of 48.23% because they enable functional evaluation of drug metabolism, transport, and toxicity responses within living cells. These assays allow researchers to analyze critical pharmacokinetic parameters such as membrane permeability, intracellular accumulation, and metabolic enzyme interactions under biologically relevant conditions.
The biochemical assays segment is expected to grow at a CAGR of 11.67%, as they provide rapid and highly specific evaluation of enzyme activity, metabolic pathways, and protein-drug interactions. These assays are increasingly used to study cytochrome P450 enzyme inhibition, metabolic stability, and drug–enzyme binding kinetics during early screening stages. Their compatibility with automated analytical platforms and high-throughput workflows allows pharmaceutical developers to generate precise mechanistic insights while supporting efficient candidate prioritization.
Application Insights
Systemic toxicity dominated the application segment with a share of 50.21% because evaluating the overall toxic effects of drug candidates across multiple organ systems is a critical requirement during preclinical development. ADME toxicology studies frequently focus on determining the influence of systematic safety profiles of drug distribution, metabolism, and accumulation before compounds advance to clinical trials.
Renal toxicity is expected to grow at a CAGR of 11.81%, largely due to the kidney’s central role in drug elimination and susceptibility to drug-induced injury. Increasing awareness of nephrotoxic risks associated with certain therapeutic classes has encouraged drug developers to incorporate early-stage kidney safety assessments within ADME testing programs, which further enhances market growth.
Competitive Landscape
The ADME toxicology testing market is moderately fragmented, with the presence of large contract research organizations (CROs), specialized toxicology testing laboratories, biotechnology service providers, and emerging technology startups. Established players typically compete on factors such as comprehensive service portfolios, regulatory compliance expertise, global laboratory networks, advanced analytical technologies, and long-standing partnerships with pharmaceutical and biotechnology companies. Emerging players tend to compete through niche capabilities, innovative in-vitro models, AI-supported predictive toxicology tools, faster turnaround times, and cost-efficient testing solutions. The market will be shaped by the growing adoption of alternative testing models, automation, and data-driven predictive toxicology approaches that improve efficiency and reduce reliance on traditional testing methods.
List of Key and Emerging Players in ADME Toxicology Testing Market
- Symeres
- Promega Corporation
- PerkinElmer Inc.
- Agilent Technologies
- Corning Inc.
- Bio-Rad Laboratories
- Covance Inc.
- Charles River Laboratories
- Eurofins Scientific
- Thermo Fisher Scientific
- GE Healthcare
- Cyprotex
- Evotec AG
- CorestemChemon
Latest News on Key and Emerging Players
| TIMELINE | COMPANY | DEVELOPMENT |
|---|---|---|
|
December 2025 |
Symeres |
Symeres obtained Science Based Targets initiative (SBTi) approval for greenhouse gas reduction goals, reflecting growing sustainability requirements from pharmaceutical clients. |
|
September 2025 |
Symeres |
Symeres acquired drug development consultancy DGr Pharma to expand integrated drug discovery and early-stage development services, including pharmacokinetic analysis, non-clinical strategy, and regulatory support related to ADME-Tox studies. |
|
July 2025 |
CorestemChemon |
CorestemChemon entered a strategic collaboration with ATG Lifetech to develop transcriptomics-based and organoid-driven preclinical testing platforms, enhancing predictive toxicology and ADME safety evaluation capabilities for pharmaceutical clients. |
Source: Secondary Research
Report Scope
| Report Metric | Details |
|---|---|
| Market Size in 2025 | USD 7.00 billion |
| Market Size in 2026 | USD 7.69 billion |
| Market Size in 2034 | USD 16.78 billion |
| CAGR | 10.24% (2026-2034) |
| Base Year for Estimation | 2025 |
| Historical Data | 2022-2024 |
| Forecast Period | 2026-2034 |
| Report Coverage | Revenue Forecast, Competitive Landscape, Growth Factors, Environment & Regulatory Landscape and Trends |
| Segments Covered | By Technology, By Testing Method, By Application |
| Geographies Covered | North America, Europe, APAC, Middle East and Africa, LATAM |
| Countries Covered | US, Canada, UK, Germany, France, Spain, Italy, Russia, Nordic, Benelux, China, Korea, Japan, India, Australia, Taiwan, South East Asia, UAE, Turkey, Saudi Arabia, South Africa, Egypt, Nigeria, Brazil, Mexico, Argentina, Chile, Colombia |
Download Free Sample Report to Get Detailed Insights.
ADME Toxicology Testing Market Segments
By Technology
- Cell Culture
- High Throughput
- Molecular Imaging
- OMICS Technology
By Testing Method
- Cellular Assay
- Biochemical Assay
- In-Silica
- Ex-vivo
By Application
- Systemic Toxicity
- Renal Toxicity
- Hepatotoxicity
- Neurotoxicity
- Other Toxicities
By Region
- North America
- Europe
- APAC
- Middle East and Africa
- LATAM
Frequently Asked Questions (FAQs)
Author's Details
Mitiksha Koul
Research Associate
Mitiksha Koul is a Research Associate with 2 years of experience in market research. She focuses on analyzing industry trends, competitive landscapes, and growth opportunities to support strategic decision-making. Mitiksha’s strong analytical skills and research expertise enable her to deliver actionable insights that help businesses adapt to evolving market dynamics and achieve sustainable growth.










