Cyclosporine Market Size, Share & Trends Analysis Report By Indication (Transplant Rejection Prophylaxis, Rheumatoid Arthritis, Psoriasis, Autoimmune Myasthenia Gravis, Dry Eye Disease, Other Indication), By Route of Administration (Oral, Parenteral) and By Region (North America, Europe, APAC, Middle East and Africa, LATAM) Forecasts, 2026-2034
Cyclosporine Market Size
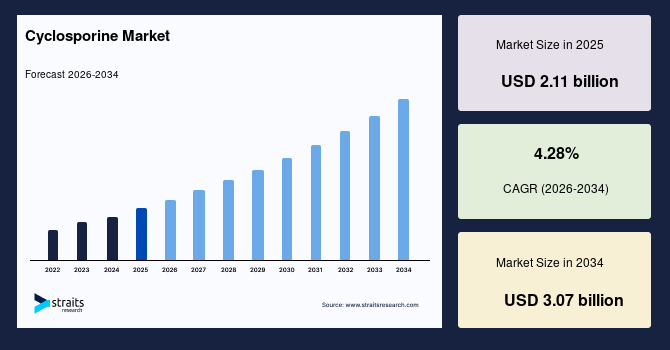
The cyclosporine market size was valued at USD 2.11 billion in 2025 and is projected to grow from USD 2.19 billion in 2026 to USD 3.07 billion by 2034 at a CAGR of 4.28% during the forecast period (2026-2034).
The cyclosporine market is growing steadily due to its essential role in organ transplantation and the treatment of autoimmune diseases such as rheumatoid arthritis and psoriasis. Rising transplant procedures and increasing prevalence of chronic immune disorders drive demand. Advances in formulation, including improved oral delivery and reduced toxicity, support better patient compliance for oral and intravenous therapies. Pharmaceutical companies focus on generics and partnerships to expand access and affordability. For example, in 2025, Novartis expanded its immunology portfolio by enhancing the distribution of its cyclosporine-based therapies in emerging markets, improving treatment accessibility. Expanding healthcare infrastructure and awareness further boost adoption. However, side effects like kidney toxicity and high long-term treatment costs remain key restraints. Ongoing research into safer immunosuppressants and personalized treatment approaches continues to shape the competitive landscape and future growth.
Key Market Insights
- North America dominated the market with the largest share of 40.63% in 2025.
- The Asia Pacific is expected to be the fastest-growing region in the market during the forecast period, registering a CAGR of 7.39%.
- Based on indication, the dry eye disease segment is expected to register a CAGR of 5.63% during the forecast period.
- Based on route of administration, the parenteral segment is projected to grow at a CAGR of 6.67% during the forecast period.
- The US cyclosporine market size was valued at USD 772.60 million in 2025 and is projected to reach USD 804.27 million in 2026.
Market Summary
| Market Metric | Details & Data (2025-2034) |
|---|---|
| 2025 Market Valuation | USD 2.11 billion |
| Estimated 2026 Value | USD 2.19 billion |
| Projected 2034 Value | USD 3.07 billion |
| CAGR (2026-2034) | 4.28% |
| Study Period | 2022-2034 |
| Dominant Region | North America |
| Fastest Growing Region | Asia Pacific |
| Key Market Players | AbbVie Inc., Strides Pharma Science Limited, Apotex Inc., EonLabs, Mayne Pharma Group Limited |
Download Free Sample Report to Get Detailed Insights.
Emerging Trends in Cyclosporine Market
Shift toward Targeted Nanocarrier & Controlled Release Delivery Platform
Shift toward nanocarrier‑based and controlled‑release delivery systems designed to improve bioavailability and reduce systemic toxicity drives market growth. Traditional formulations of cyclosporine are hampered by poor solubility and a narrow therapeutic index, prompting R&D into microemulsion, lipid nanoparticles, and sustained‑release implant technologies. By enhancing penetration and maintaining stable drug levels, these platforms improve therapeutic outcomes and patient adherence, differentiating products beyond generics.
Inclination toward Water‑free & Nanomicellar Eye Drops
The growing adoption of advanced ophthalmic formulations designed specifically for chronic ocular surface disorders like dry eye disease is another trend expanding market growth. Newer water-free and nanomicellar eye drops improve drug solubility and enable deeper corneal penetration while minimizing irritation and dosing frequency compared to traditional emulsions. For example, nanomicellar cyclosporine formulations have shown significantly higher drug retention in tear film layers, leading to better patient compliance. This segment continues to expand as ophthalmologists increasingly prefer localized immunomodulation over systemic therapies for long-term disease management.
Market Drivers
Integration of Low-dose Cyclosporine in Combination Protocols and Autoimmune Indications Drives Market
The increasing integration of low-dose cyclosporine into combination immunosuppressive protocols is a key market driver, particularly in organ transplantation. Clinicians combine cyclosporine with agents such as mycophenolate mofetil and corticosteroids to balance efficacy and reduce nephrotoxicity while maintaining graft survival. A 2025 clinical study in kidney transplant recipients shows that over 80% of patients maintained therapeutic levels under structured protocols, supporting optimized dosing strategies. With more than 48,000 organ transplants performed annually in the US, demand for such combination regimens continues to expand significantly.
Growing use of cyclosporine in steroid-refractory autoimmune conditions drives market growth. Clinicians increasingly prescribe cyclosporine in diseases such as severe aplastic anemia and refractory uveitis due to its rapid immunosuppressive action. A 2025 clinical study in patients with refractory uveitis reported that nearly 70% achieved significant inflammation control within three months of initiation. This targeted use in difficult-to-treat patient subsets strengthens its demand in specialty care settings, where alternative treatment options remain limited.
Market Restraints
High Interpatient Pharmacokinetic Variability and Stringent Regulatory Scrutiny on Bioequivalence Restrain Market Growth
A key restraint in the market is its significant interpatient pharmacokinetic variability, which complicates standardized dosing. Factors such as genetic polymorphisms in CYP3A enzymes, food interactions, and variable absorption rates lead to unpredictable drug exposure levels. Clinicians must frequently adjust doses through intensive monitoring, increasing treatment complexity and healthcare burden. This variability limits the adoption of cyclosporine in less specialized settings where monitoring infrastructure is limited. As newer immunosuppressants with more predictable profiles emerge, this slows their market expansion.
The strict regulatory requirements for bioequivalence of generic formulations due to their narrow therapeutic index restrain market growth. For example, the European Medicines Agency (EMA) mandates tighter 90-111% bioequivalence limits for narrow therapeutic index drugs, increasing generic approval complexity and limiting product substitution. Thus, small variations in drug absorption significantly impact efficacy and toxicity, leading regulators to enforce rigorous clinical testing and post-approval monitoring. This increases development costs and delays market entry for generic manufacturers. This limits generic substitution rates, restricts price competition, and creates barriers for new entrants, ultimately constraining market growth despite cyclosporine’s long-standing presence.
Market Opportunities
Development of Topical Formulations and Bundled Transplant Care Supply Agreements Offer Growth Opportunities for Market Players
The development of ultra-low-dose topical formulations for localized dermatological conditions such as chronic hand eczema and limited psoriasis. These formulations enable targeted delivery directly to affected skin while minimizing systemic toxicity risks. For example, the growing off-label dermatological use of oral brands like Sandimmune is being reformulated by compounding pharmacies into topical preparations for localized lesions. This demand signals a clear commercial gap, encouraging companies to develop standardized topical products for safer, long-term dermatology applications.
The expansion of bundled procurement agreements with transplant hospital networks offers growth opportunities for market player. Cyclosporine is supplied as part of standardized post-transplant immunosuppressive kits alongside adjunct therapies. This model shifts competition from standalone drug pricing to integrated care solutions, including adherence monitoring support and dosing optimization services aligned with transplant protocols. For example, the increasing use of United Network for Organ Sharing (UNOS)-linked transplant centers in the US, where bundled immunosuppression pathways are being standardized across kidney and liver transplant programs to improve outcomes and control long-term treatment costs.
Regional Insights
North America: Market Leadership Driven by Strong Post-Transplant Immunosuppression Protocols and Increasing Prevalence of Psoriasis
North America market accounted for a revenue share of 40.63% in 2025 due to highly structured transplant allocation systems and strong post-transplant immunosuppression protocols in the US, where hospitals routinely use standardized cyclosporine-based regimens in kidney and liver transplants. Widespread insurance coverage through Medicare and private payers supports long-term immunosuppressant adherence after surgery. For example, large transplant centers like Mayo Clinic and Cleveland Clinic maintain protocol-driven use. High ophthalmic cyclosporine adoption for dry eye treatment supports regional demand. These factors collectively drive market growth in North America.
The US market is growing due to highly protocol-driven transplant care pathways and strong integration of immunosuppressant management within large hospital networks. Major transplant centers such as Mayo Clinic and Cleveland Clinic use standardized post-kidney and liver transplant regimens where cyclosporine remains part of long-term maintenance therapy. Highly developed pharmacy-led therapeutic drug monitoring (TDM) services integrated within hospital electronic health record systems, which support precise dosing of narrow therapeutic index immunosuppressants.
The Canadian market is driven by province-level biosimilar switching mandates and hospital formulary harmonization policies, which indirectly strengthen branded and originator cyclosporine utilization in stable transplant patients where switching is clinically restricted due to narrow therapeutic index concerns. For example, Ontario Health and British Columbia Pharmacare maintain strict interchangeability rules for immunosuppressants, requiring physician justification before switching stable post-transplant patients. Growing cases of Psoriasis, especially moderate-to-severe plaque psoriasis, drive market demand during flare control phases in dermatology clinics. These factors are driving the market growth in Canada.
Asia Pacific: Fastest Growth Driven by Rising Prevalence of Autoimmune Conditions and Increasing Domestic Manufacturing of Immunosuppressants
The Asia Pacific cyclosporine market is expected to register the fastest growth with a CAGR of 7.39% during the forecast period, due to high autoimmune disease burden, expanding transplant infrastructure, and rapid ophthalmic drug adoption in emerging economies. Rising prevalence of rheumatoid arthritis and Myasthenia Gravis in countries like India, China, and Japan, where increasing urbanization and changing lifestyles are contributing to higher chronic inflammation cases. For example, large tertiary hospitals in India, such as AIIMS New Delhi, report growing use of cyclosporine as a second-line therapy in steroid-resistant rheumatoid arthritis and severe psoriasis cases. High ophthalmic disease burden, especially dry eye linked to digital screen exposure in Japan and South Korea, is driving strong adoption of cyclosporine eye drops in specialist eye clinics and retail pharmacy channels.
The Chinese market is growing due to the rapid expansion of domestic generic immunosuppressant manufacturing under national volume-based procurement programs, which improves affordability and hospital adoption. Strong growth in hospital-led organ transplant registries also increases standardized post-transplant cyclosporine use in kidney transplant centers in Beijing and Shanghai. Rising integration of Traditional Chinese Medicine hospitals with Western immunosuppressive therapy pathways supports combination treatment adoption. For example, large tertiary hospitals like Peking Union Medical College Hospital increasingly follow protocol-based use in complex autoimmune and transplant cases.
The Japanese market is driven by a high burden of ophthalmic and autoimmune disorders combined with advanced healthcare access. In Japan, dry eye disease affects nearly 22 million people, largely due to an aging population and prolonged digital device usage, which significantly increases demand for cyclosporine-based eye drops in ophthalmic clinics and retail pharmacies. Strong domestic generic manufacturing ecosystem concentrated in hospital injectables and off-patent immunosuppressants, which ensures stable local supply and cost efficiency for transplant care. Leading Japanese manufacturers such as Nichi-Iko Pharmaceutical, Sawai Pharmaceutical, and Towa Pharmaceutical play a key role in supplying hospital-based formulations under strict quality standards.
By Indication
The transplant rejection prophylaxis segment dominated the market, with a revenue share of 31.70% in 2025, due to the critical need for lifelong immunosuppression in organ transplantation. For example, over 100,000 people in the US live with functioning organ transplants, all requiring continuous therapy like cyclosporine. Kidney transplants and standardized post-transplant protocols in major hospitals further sustain a consistent segment.
The dry eye disease segment is expected to grow at a CAGR of 5.63% during the forecast period, due to increasing adoption of post-refractive surgery and cataract care protocols that routinely include anti-inflammatory therapy to manage ocular surface inflammation. For example, LASIK and cataract surgery follow-up care in large ophthalmology centers increasingly prescribes cyclosporine eye drops to improve tear film recovery and reduce chronic dryness symptoms. Expanding elderly populations with age-related meibomian gland dysfunction further increases demand for such treatments.
By Route of Administration
The oral segment is expected to grow at a CAGR of 5.84% during the forecast period, due to high patient convenience and strong outpatient compatibility, as it avoids hospital-based administration and allows long-term self-managed immunosuppression. Oral cyclosporine also provides flexible dose titration, which is critical in transplant and autoimmune care where therapeutic drug levels must be frequently adjusted. Oral route also ensures steady systemic absorption for chronic maintenance therapy, making it preferred over injectables for long-term use. These factors collectively drive segment growth.
The parenteral segment is expected to grow at a CAGR of 6.67% during the forecast period due to its rapid onset of action and immediate bioavailability, making it critical in acute transplant rejection episodes where fast immunosuppression is required. It is also preferred in hospital and ICU settings for patients unable to tolerate oral drugs. Parenteral cyclosporine ensures precise dose control during initial stabilization phases, reducing early rejection risk.
Competitive Landscape
The cyclosporine market is moderately consolidated with a mix of global innovators and strong generic manufacturers competing across transplant, autoimmune, and ophthalmic segments. Leading players such as Novartis, AbbVie, Sandoz, Viatris, Teva, Sun Pharmaceutical Industries, Cipla, Apotex, Lupin, and Dr. Reddy’s dominate through broad portfolios, strong regulatory compliance, and extensive distribution networks. Competition is intensified by price erosion from generics and frequent formulation upgrades, especially in ophthalmic cyclosporine. Companies focus on bioequivalence optimization, novel delivery systems, and lifecycle management strategies to defend market share. Strategic partnerships, regional expansion, and hospital supply agreements further shape competitive positioning globally.
List of Key and Emerging Players in Cyclosporine Market
- AbbVie Inc.
- Strides Pharma Science Limited
- Apotex Inc.
- EonLabs
- Mayne Pharma Group Limited
- Intas Pharmaceuticals Ltd.
- Novartis AG
- Santen Pharmaceutical Co., Ltd.
- Sun Pharmaceutical Industries Ltd.
- Teva Pharmaceutical Industries Ltd.
- Viatris Inc.
- Cipla Ltd.
- Reddy’s Laboratories Ltd.
- Glenmark Pharmaceuticals Ltd.
- Pfizer Inc.
- Astellas Pharma Inc.
Recent Developments
- In December 2025, Amneal Pharmaceuticals, Inc. received US FDA approval for cyclosporine ophthalmic emulsion 0.05%, a generic version of Restasis used for dry eye disease.
- In January 2026, Bora Pharmaceuticals Co., Ltd. announced US FDA approval for its cyclosporine ophthalmic emulsion 0.05%.
Report Scope
| Report Metric | Details |
|---|---|
| Market Size in 2025 | USD 2.11 billion |
| Market Size in 2026 | USD 2.19 billion |
| Market Size in 2034 | USD 3.07 billion |
| CAGR | 4.28% (2026-2034) |
| Base Year for Estimation | 2025 |
| Historical Data | 2022-2024 |
| Forecast Period | 2026-2034 |
| Report Coverage | Revenue Forecast, Competitive Landscape, Growth Factors, Environment & Regulatory Landscape and Trends |
| Segments Covered | By Indication, By Route of Administration |
| Geographies Covered | North America, Europe, APAC, Middle East and Africa, LATAM |
| Countries Covered | US, Canada, UK, Germany, France, Spain, Italy, Russia, Nordic, Benelux, China, Korea, Japan, India, Australia, Taiwan, South East Asia, UAE, Turkey, Saudi Arabia, South Africa, Egypt, Nigeria, Brazil, Mexico, Argentina, Chile, Colombia |
Download Free Sample Report to Get Detailed Insights.
Cyclosporine Market Segments
By Indication
- Transplant Rejection Prophylaxis
- Rheumatoid Arthritis
- Psoriasis
- Autoimmune Myasthenia Gravis
- Dry Eye Disease
- Other Indication
By Route of Administration
- Oral
- Parenteral
By Region
- North America
- Europe
- APAC
- Middle East and Africa
- LATAM
Frequently Asked Questions (FAQs)
Author's Details
Dhanashri B
Senior Research Associate
Dhanashri Bhapakar is a Senior Research Associate with 3+ years of experience in the Biotechnology sector. She focuses on tracking innovation trends, R&D breakthroughs, and market opportunities within biopharmaceuticals and life sciences. Dhanashri’s deep industry knowledge enables her to provide precise, data-backed insights that help companies innovate and compete effectively in global biotech markets.










