Molecular Biology Enzymes, Kits, and Reagents Market Size, Share & Trends Analysis Report By Product (Kits & Reagents, Enzymes, Reverse Transcriptase, Ligases & Kinases, Nucleases & Restriction Enzymes, Modifying Enzymes, Other Specialty Enzymes), By Application (Polymerase Chain Reaction, Next-generation Sequencing, Epigenetics, Cloning & Synthetic Biology, Gene-Editing /CRISPR Workflows, Protein Analysis & Proteomics, Other Applications), By End User (Hospitals & Clinics, Diagnostic Centers, Academic & Research Institutes, Pharmaceutical & Biotechnology Companies, Contract Research & Manufacturing Organizations, Other End Users) and By Region (North America, Europe, APAC, Middle East and Africa, LATAM) Forecasts, 2026-2034
What is the size of Molecular Biology Enzymes, Kits, and Reagents Market?
The molecular biology enzymes, kits, and reagents market size was valued at USD 15.42 billion in 2025 and is projected to grow from USD 16.97 billion in 2026 to USD 36.99 billion by 2034 (2026-2034), growing at a CAGR of 10.23% during the forecast period.
Key Market Insights
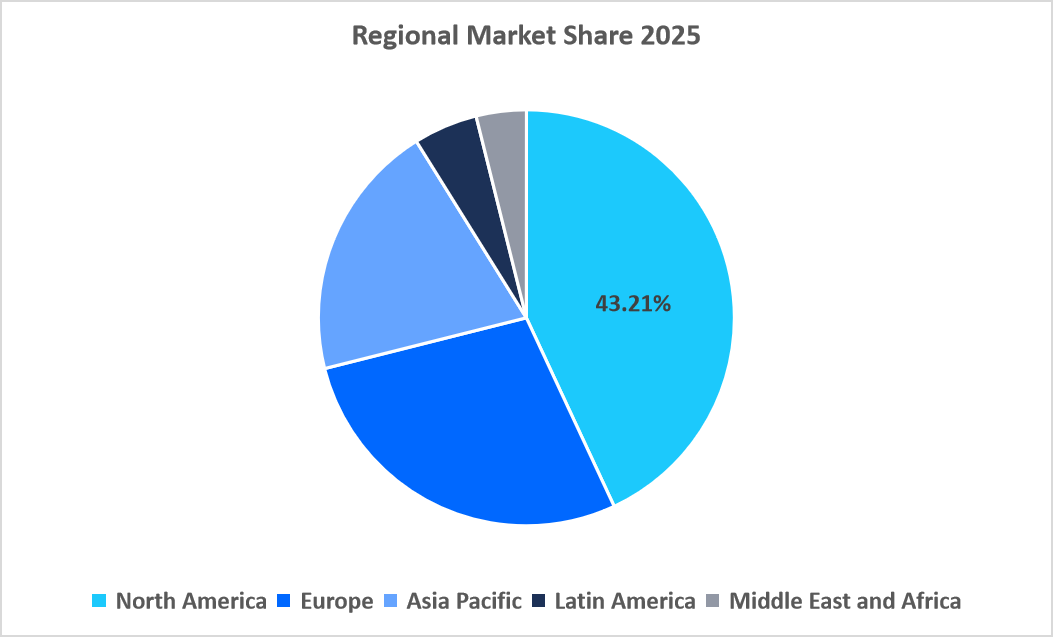
- North America dominated the molecular biology enzymes, kits, and reagents market, with the largest share of 43.21% in 2025.
- The Asia Pacific is expected to be the fastest-growing region in the molecular biology enzymes, kits, and reagents market during the forecast period at a CAGR of 12.64%.
- By product, the kits & reagents segment dominated the market with the largest share in 2025.
- By application, the polymerase chain reaction segment is projected to grow at a considerable CAGR of 11.45% during the forecast period.
- By end user, the pharmaceutical & biotechnology companies segment dominated the market with the largest share of 36.44% in 2025.
- The US molecular biology enzymes, kits, and reagents market size was valued at USD 5.51 billion in 2025 and is expected to reach USD 6.07 billion in 2026.
Market Summary
| Market Metric | Details & Data (2025-2034) |
|---|---|
| 2025 Market Valuation | USD 15.42 billion |
| Estimated 2026 Value | USD 16.97 billion |
| Projected 2034 Value | USD 36.99 billion |
| CAGR (2026-2034) | 10.23% |
| Study Period | 2022-2034 |
| Dominant Region | North America |
| Fastest Growing Region | Asia-Pacific |
| Key Market Players | Thermo Fisher Scientific Inc., QIAGEN N.V., Merck KGaA, New England Biolabs, Inc., Agilent Technologies, Inc. |

Download Free Sample Report to Get Detailed Insights.
What are the Top Trends in Molecular Biology Enzymes, Kits, and Reagents Market?
The launch of high-fidelity and specialized enzymes is a key market trend, as manufacturers develop products optimized for next-generation sequencing and gene editing workflows. These enzymes improve accuracy, reduce error rates, and support advanced molecular applications, strengthening adoption in research and clinical laboratories.
The increasing focus on environmental sustainability is a major market shift, as life science companies incline toward eco-friendly reagents and greener manufacturing practices. This includes reduced hazardous chemicals, recyclable packaging, and lower energy consumption, aligning reagent portfolios with the sustainability goals of research institutions.
The rising use of CRISPR technologies is a key market trend, as more companies offer engineered nucleases and ready-to-use editing kits. Integrated DNA Technologies offers CRISPR-Cas systems and guide RNA solutions that support genome editing.
The integration of bioinformatics and digital workflow support results in reagent suppliers bundling enzymes and kits with protocol optimization tools and cloud-based platforms. Companies offer sequencing reagents alongside software solutions that improve workflow efficiency, data analysis, and reproducibility.
What are the Driving Factors in Molecular Biology Enzymes, Kits, and Reagents Market?
Rising number of approved cell and gene therapies increases demand for specialized molecular biology enzymes. These therapies require enzymes for plasmid preparation, nucleic acid amplification, and viral vector construction, which raises demand for GMP-grade and regulatory-compliant reagents.
Pharmaceutical and biotechnology companies expand mRNA production capacity to support large-scale commercial vaccine supply, which increases consumption of enzymes for transcription, capping, purification, and quality control. This drives sustained demand growth for high-volume and high-purity molecular biology reagents across manufacturing supply chains.
Greater reliance on biomarker-driven treatment decisions raises the use of PCR enzymes, reverse transcriptases, and library preparation kits in clinical testing workflows. This leads to higher recurring demand from diagnostic laboratories and supports stable reagent consumption.
New biologics and vaccine facilities require enzymes and kits for quality control, validation, and regulatory testing. This strengthens regional supply chains and supports long-term growth in demand for molecular biology reagents in emerging manufacturing hubs.
Which Factors Restraints the Growth of Molecular Biology Enzymes, Kits, and Reagents Market?
Most molecular biology enzymes require strict temperature control to maintain stability and activity, which increases logistics complexity and distribution costs. High-grade polymerase requires high operational temperatures and loses functionality if storage conditions are not maintained. This limits adoption in cost-sensitive and infrastructure-constrained regions, thereby slowing overall market expansion.
Patents and licensing requirements for modified enzymes and CRISPR technologies raise development and commercialization costs for smaller players. Licensing genome-editing tools results in increased upfront expenses for new entrants, which restricts competition and slows the pace of innovation-driven adoption.
Export controls and regulatory barriers on advanced biotechnology materials disrupt cross-border sourcing of enzymes, reagents, and raw materials. These disruptions increase lead times and procurement uncertainty, which constrains consistent supply availability and hinders the molecular biology enzymes, kits, and reagents market growth.
What are the Growth Opportunities in Molecular Biology Enzymes, Kits, and Reagents Market?
Increasing adoption of automated liquid handling systems acts as a growth factor for the market. Manufacturers integrate pre-aliquoted reagent kits and sample-to-result platforms that combine extraction and detection reagents to improve workflow efficiency and reduce handling errors. This creates a growth avenue for players by enabling premium, automation-ready product offerings and deeper penetration into high-throughput laboratory environments.
Rising outsourcing of reagent production by diagnostic companies and biotech startups offers growth opportunities. Established reagent manufacturers integrate contract development and private-label production capabilities to support external product pipelines. This helps generate stable B2B revenue streams and long-term supply partnerships in the molecular biology enzymes, kits, and reagents market.
Expanding use of molecular biology reagents in veterinary and agricultural genomics is expected to drive market growth. Suppliers integrate specialized enzymes, kits, and assays tailored for animal health, breeding, and crop genomics applications. This offers lucrative growth opportunities for market players by diversifying end-user markets and reducing dependence on human healthcare research.
Regional Analysis
North America Molecular Biology Enzymes, Kits, and Reagents Market
The molecular biology enzymes, kits, and reagents market in North America had a market share of 43.21% in 2025. The growth is facilitated by highly concentrated regional innovation hubs and genomics ecosystems. The region boasts a high number of biotech companies, which drives high demand for enzymes and reagents in innovative research. Academic institutions, research hospitals, and national laboratories conduct large-scale sequencing and functional genomics studies, which drive recurring demand for library preparation kits, polymerases, reverse transcriptase, and nucleic acid purification reagents. Public research funding, venture capital investment, and industry-sponsored R&D provide stable financial support for life science research, ensuring consistent procurement of molecular biology reagents despite economic cycles. High penetration of molecular diagnostics in infectious disease testing, oncology, and genetic screening increases the routine use of PCR and amplification reagents.
Source: Straits Research
Asia Pacific is expected to be the fastest-growing region, growing at a CAGR of 12.64% from 2026 to 2034. This growth is augmented by the rapid expansion of regional biotech and genomics research capacity, with countries such as China, India, Japan, and South Korea showing an increase in the number of molecular labs and the number of tests performed. The pharmaceutical and biotech industry is shifting to local reagent manufacturers to cut costs and increase adoption.
Europe Molecular Biology Enzymes, Kits, and Reagents Market
The European market is experiencing strong growth due to strong public research funding, such as Horizon Europe, which allocates investment into health research and genomic studies, driving reagent demand. Governments in this region allocate substantial resources to life science projects that boost reagent demand. The region has a growing pipeline of advanced therapies moving from clinical trials to commercial manufacturing, which increases demand for high-purity, regulatory-compliant enzymes used in vector production, quality control, and validation workflows.
Latin America Molecular Biology Enzymes, Kits, and Reagents Market
Latin America is increasingly selected for multinational clinical trials due to patient availability and cost efficiency. Clinical research organizations and trial sites increase procurement of molecular kits for biomarker testing, genetic screening, and sample analysis. Brazil and Mexico are focusing on infrastructure development to improve molecular testing, offering new growth opportunities.
Middle East & Africa Molecular Biology Enzymes, Kits, and Reagents Market
The Middle East & Africa market growth is propelled by rising government efforts to promote genomic research and diagnostic capabilities in healthcare. The region is focusing on genomic surveillance for infectious diseases and endemic diseases such as HIV and TB. Spending on regional reference labs, biobanks, and multi-omics research centers is driving reagent use, thereby supporting market growth.
Product Insights
The kits & reagents segment is expected to register a CAGR of 11.56% during the forecast period, driven by increasing demand for workflow-based, ready-to-use solutions such as PCR Master Mix & qPCR kits, NGS/library preparation kits, and DNA/RNA extraction & purification kits. The rising adoption of automation in diagnostic laboratories and expanding genomic research activities are accelerating demand for standardized and contamination-controlled kits.
The enzymes segment is expected to register a growth rate of 11.16% during the forecast period. Growth is supported by continuous innovation in DNA polymerases (including high-fidelity, thermostable, and strand-displacing variants), reverse transcriptases, ligases & kinases, and restriction enzymes. Expanding applications in gene editing, synthetic biology, and high-precision sequencing workflows are increasing the demand for engineered enzymes with enhanced specificity and efficiency.
Application Insights
Next-Generation Sequencing (NGS) dominated the application segment with a share of 34.51% in 2025. The segment’s large share is attributed to increasing use in oncology research, rare disease diagnostics, reproductive health screening, and precision medicine initiatives. Pharmaceutical and biotechnology companies are significantly investing in sequencing-based biomarker discovery and drug development, strengthening NGS demand for specialized enzymes and library preparation reagents.
The polymerase chain reaction (PCR) is expected to register a CAGR of 11.45% during the forecast period. The segment continues to maintain steady demand due to its critical role in clinical diagnostics, infectious disease detection, and research applications. Continuous advancements in real-time PCR and multiplex PCR technologies are supporting consistent reagent and enzyme consumption.
End User Insights
Pharmaceutical & biotechnology companies held the largest share of the end-user segment at 36.44% in 2025. Their dominance is driven by extensive use of molecular biology enzymes and kits in drug discovery, clinical trials, biologics development, and precision medicine research. Growing R&D expenditure and increasing focus on genomics-driven therapeutics further reinforce this segment’s leadership.
The academic & research institutes segment is expected to register higher growth with a CAGR of 11.63% during the forecast period. Growth is fueled by expanding research grants, government-funded genomics initiatives, and a rising focus on CRISPR gene editing, epigenetics, and synthetic biology research. Increasing collaboration between academia and industry is also contributing to higher adoption of molecular biology reagents and enzymes.
Competitive Landscape
The molecular biology enzymes, kits, and reagents market is moderately consolidated, dominated by established players such as Thermo Fisher Scientific, Merck KGaA, QIAGEN, and New England Biolabs, which maintain strong market positions through their product portfolios, strong distribution networks, and continuous innovation in kit technology. Emerging companies compete by offering specialized enzymes, cost-effective kits & reagents for research applications within domestic networks. Specialty reagent and enzyme innovators focus on niche or advanced products for technical differentiation and cutting-edge workflows. Contract manufacturing and private-label producers compete on OEM partnerships and the ability to scale with demand fluctuations.
List of Key and Emerging Players in Molecular Biology Enzymes, Kits, and Reagents Market
- Thermo Fisher Scientific Inc.
- QIAGEN N.V.
- Merck KGaA
- New England Biolabs, Inc.
- Agilent Technologies, Inc.
- Promega Corporation
- Hoffmann-La Roche Ltd.
- Bio-Rad Laboratories, Inc.
- Takara Bio Inc.
- Illumina, Inc.
- Danaher Corporation
- PerkinElmer, Inc.
- Integrated DNA Technologies, Inc.
- Enzo Life Sciences, Inc.
- Abcam plc
- Meridian Bioscience, Inc.
- PCR Biosystems
- Omega Bio-tek, Inc.
- Others
Latest News on Key and Emerging Players
|
TIMELINE |
COMPANY |
DEVELOPMENT |
|---|---|---|
|
February 2026 |
Promega Corporation |
The National Medical Products Administration (NMPA) approved Promega’s OncoMate Microsatellite Instability (MSI) Detection Kit. |
|
January 2026 |
Illumina, Inc. |
Illumina acquired SomaLogic, a leading developer of proteomics technology. |
|
November 2025 |
PCR Biosystems |
PCR Biosystems launched its new Lyo-Ready Inhibitor-Tolerant reagents for molecular diagnostics. |
|
November 2025 |
New England Biolabs |
New England Biolabs and Volta Labs partnered to expand the portfolio of NEBNext reagents available on the Callisto Sample Preparation Platform. |
|
July 2025 |
New England Biolabs |
The company launched its NEBNext Low-Bias Small RNA Library Prep Kit to reduce biased representation of small RNA species in sequencing data. |
Source: Secondary Research
Report Scope
| Report Metric | Details |
|---|---|
| Market Size in 2025 | USD 15.42 billion |
| Market Size in 2026 | USD 16.97 billion |
| Market Size in 2034 | USD 36.99 billion |
| CAGR | 10.23% (2026-2034) |
| Base Year for Estimation | 2025 |
| Historical Data | 2022-2024 |
| Forecast Period | 2026-2034 |
| Report Coverage | Revenue Forecast, Competitive Landscape, Growth Factors, Environment & Regulatory Landscape and Trends |
| Segments Covered | By Product, By Application, By End User |
| Geographies Covered | North America, Europe, APAC, Middle East and Africa, LATAM |
| Countries Covered | US, Canada, UK, Germany, France, Spain, Italy, Russia, Nordic, Benelux, China, Korea, Japan, India, Australia, Singapore, Taiwan, South East Asia, UAE, Turkey, Saudi Arabia, South Africa, Egypt, Nigeria, Brazil, Mexico, Argentina, Chile, Colombia |
Download Free Sample Report to Get Detailed Insights.
Molecular Biology Enzymes, Kits, and Reagents Market Segments
By Product
-
Kits & Reagents
- PCR Master-Mix & qPCR Kits
- NGS/Library Preparation Kits
- DNA/RNA Extraction & Purification Kits
- Lyophilized Field-Use Kits
- Purification Kits
- Others
-
Enzymes
-
DNA Polymerases
- High Fidelity DNA Polymerases
- Thermostable DNA Polymerases
- Strand-displacing DNA Polymerases
-
DNA Polymerases
- Reverse Transcriptase
- Ligases & Kinases
- Nucleases & Restriction Enzymes
- Modifying Enzymes
- Other Specialty Enzymes
By Application
- Polymerase Chain Reaction
- Next-generation Sequencing
- Epigenetics
- Cloning & Synthetic Biology
- Gene-Editing /CRISPR Workflows
- Protein Analysis & Proteomics
- Other Applications
By End User
- Hospitals & Clinics
- Diagnostic Centers
- Academic & Research Institutes
- Pharmaceutical & Biotechnology Companies
- Contract Research & Manufacturing Organizations
- Other End Users
By Region
- North America
- Europe
- APAC
- Middle East and Africa
- LATAM
Frequently Asked Questions (FAQs)
Author's Details
Debashree B
Healthcare Lead
Debashree Bora is a Healthcare Lead with over 7 years of industry experience, specializing in Healthcare IT. She provides comprehensive market insights on digital health, electronic medical records, telehealth, and healthcare analytics. Debashree’s research supports organizations in adopting technology-driven healthcare solutions, improving patient care, and achieving operational efficiency in a rapidly transforming healthcare ecosystem.










