Allergy Diagnostics and Therapeutics Market Size, Share & Trends Analysis Report By Type (Diagnostics, Therapeutics), By Allergen Type (Food, Inhaled, Drug, Other Allergen Types), By Test Type (In Vivo Tests, In Vitro Tests) and By Region (North America, Europe, APAC, Middle East and Africa, LATAM) Forecasts, 2026-2034
What is the Size of Allergy Diagnostics and Therapeutics Market?
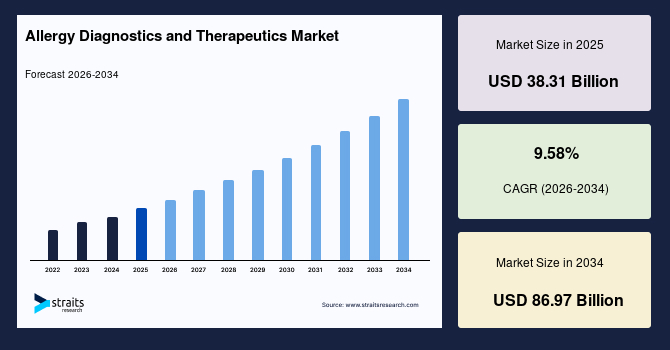
The allergy diagnostics and therapeutics market size was valued at USD 37.03 billion in 2025 and is projected to grow from USD 40.60 billion in 2026 to USD 85.94 billion by 2034 at a CAGR of 9.83% during the forecast period (2026-2034).
Key Market Insights
- North America dominated the allergy diagnostics and therapeutics market with the largest share of 37.62% in 2025.
- The Asia Pacific is expected to be the fastest-growing region in the allergy diagnostics and therapeutics market during the forecast period, registering a CAGR of 11.64%.
- Based on type, the diagnostics segment is expected to register a CAGR of 10.37% during the forecast period.
- Based on allergen type, the inhaled segment dominated the market with a revenue share of 48.50% in 2025.
- By test type, the in vivo tests segment is projected to grow at a CAGR of 10.42% during 2026-2034.
- The US allergy diagnostics and therapeutics market size was valued at USD 12.53 billion in 2025 and is projected to reach USD 13.74 billion in 2026.
Market Summary
| Market Metric | Details & Data (2025-2034) |
|---|---|
| 2025 Market Valuation | USD 37.03 billion |
| Estimated 2026 Value | USD 40.60 billion |
| Projected 2034 Value | USD 85.94 billion |
| CAGR (2026-2034) | 9.83% |
| Study Period | 2022-2034 |
| Dominant Region | North America |
| Fastest Growing Region | Asia Pacific |
| Key Market Players | Thermo Fisher Scientific, Inc., DASIT Group SPA, AESKU.GROUP GmbH, bioMérieux, Lilly |
Download Free Sample Report to Get Detailed Insights.
What are the Latest Trends in Allergy Diagnostics and Therapeutics Market?
Clinical laboratories are increasingly adopting component-resolved diagnostics (CRD) to identify specific allergenic proteins instead of whole allergen extracts. This approach helps distinguish true allergies from cross-reactivity in complex cases, while multiplex microarray platforms enable simultaneous testing of multiple allergen components, improving diagnostic precision and immunotherapy decisions.
A new generation of recombinant and peptide-based allergen vaccines is emerging as a safer alternative to traditional allergen extract immunotherapy. By modifying IgE-binding regions while preserving immune-activating components, these vaccines reduce allergic reactions and enable more standardized, consistent, and potentially shorter immunotherapy treatments.
Use of nanotechnology-based allergen delivery systems in immunotherapy by encapsulating allergens in biodegradable nanoparticles or liposomes. These delivery systems allow controlled release of allergenic peptides and targeted uptake by antigen-presenting cells, reducing systemic exposure and minimizing adverse reactions. Preclinical and early clinical studies suggest this approach shortens treatment durations and improves patient adherence compared with traditional multi-year immunotherapy regimens.
What are the Key Drivers in Allergy Diagnostics and Therapeutics Market?
The increasing adoption of biologics targeting immune mediators like IgE, IL-4, IL-5, and IL-13 allows clinicians to provide more precise treatment for severe allergic diseases. This drives demand for specialized therapeutic antibodies and supports manufacturers in scaling production of targeted biologics.
Allergy clinics are increasingly using basophil activation tests to measure immune cell responses in drug, food, and venom allergies. This improves diagnostic accuracy and reduces reliance on risky oral challenge tests, increasing demand for functional diagnostic assays and related laboratory reagents. Consequently, suppliers of allergy testing platforms and consumables experience higher adoption and growth in the diagnostics segment.
Laboratories are implementing multiplex molecular panels that detect multiple allergen components in a single assay. This enhances precision by distinguishing cross-reactive sensitizations from true allergies, increasing demand for advanced diagnostic kits and equipment. The outcome is broader uptake of personalized immunotherapy planning tools, boosting market supply and driving growth in the allergy diagnostics sector.
What are the Restraining Factors in Allergy Diagnostics and Therapeutics Market?
Allergen cross-reactivity between botanically or structurally related allergens complicates the interpretation of conventional IgE tests. Misidentification of the primary allergen source lead to inaccurate diagnosis or immunotherapy selection, reducing treatment effectiveness and slowing clinician confidence in certain diagnostic methods.
Next-generation allergy diagnostic tools, including molecular allergen assays and cellular activation tests, face limited large-scale clinical validation. The absence of standardized reference ranges and long-term outcome data restricts widespread adoption by hospitals and primary care providers.
The risk of systemic allergic reactions during allergen immunotherapy during dose-escalation phases remains a significant restraint in the market. This safety concern requires close medical supervision and controlled clinical settings, limiting treatment scalability, reducing patient acceptance, and slowing broader adoption in routine outpatient care.
What are the Growth Opportunities for Players in Allergy Diagnostics and Therapeutics Market?
Improvements in mRNA platforms enable the development of next-generation allergy vaccines that encode specific allergen proteins. This creates growth opportunities for pharmaceutical companies to design faster, more precise immunotherapies that shorten treatment durations and improve immune tolerance.
Nasal sprays and topical formulations containing allergen-binding molecules are gaining research interest for blocking allergens before they trigger immune responses. This opens opportunities for companies to develop preventive therapies for allergic rhinitis and other airborne allergen-related conditions. Barrier therapies have become a widely adopted first-line preventive solution, reducing reliance on long-term pharmacological treatments.
Therapies using short allergen fragments target specific immune pathways to induce tolerance while minimizing IgE-mediated reactions. This presents growth potential for safer, highly targeted treatments for respiratory and food allergies.
Regional Analysis
North America Allergy Diagnostics and Therapeutics Market
North America held a dominating share of the market in 2025 with a share of 37.62% due to the strong presence of specialized allergy treatment infrastructure and clinical research activities. The US has more than 1,200 dedicated allergy clinics providing immunotherapy, treating more than 4.5 million patients annually, which significantly accelerates the adoption of diagnostic tests and targeted therapeutics. Rapid integration of electronic health record–linked allergy decision-support systems in hospitals automatically flag potential drug or environmental allergies during prescribing, prompting confirmatory diagnostic testing and specialist referrals. These factors collectively drive market growth in North America.
Asia Pacific Allergy Diagnostics and Therapeutics Market
The Asia Pacific allergy diagnostics and therapeutics market is anticipated to register a CAGR of 11.64% during the forecast period. The market is growing due to the rapid expansion of large-scale national allergy screening programs and hospital network modernization. China’s National Health Commission has launched programs targeting the screening of nearly 50 million school children for allergic diseases, significantly increasing demand for diagnostic assays and follow-up therapies. Cities such as Beijing, Delhi, and Seoul report rising allergy prevalence due to particulate pollution and urban environmental exposure, expanding allergy testing programs and respiratory allergy treatment services.
Europe Allergy Diagnostics and Therapeutics Market
The allergy diagnostics and therapeutics market in Europe is supported by strong immunotherapy manufacturers and standardized allergen production facilities across countries such as Germany, France, and Spain. These facilities manufacture pollen and mite allergen extracts, enabling precise immunotherapy formulations tailored to Europe’s diverse environmental allergen profiles. The implementation of pan-European allergy surveillance and research initiatives, such as multicenter clinical networks that track allergen prevalence and treatment outcomes. Such initiatives further boost market growth in European countries.
Middle East & Africa Allergy Diagnostics and Therapeutics Market
The Middle East & Africa allergy diagnostics and therapeutics market is primarily driven by the development of national allergy registries and structured surveillance programs in countries such as the UAE. The UAE’s National Allergy Registry collects epidemiological and treatment outcome data, enabling standardized diagnostic protocols and improving access to allergy testing. Increasing import and distribution partnerships for advanced allergy biologics and diagnostic platforms further support market growth.
Latin America Allergy Diagnostics and Therapeutics Market
The allergy diagnostics and therapeutics market in Latin America is supported by rapid expansion of allergy testing volumes in Brazil’s private healthcare networks, where approximately 5.1 million allergy diagnostic tests are performed annually, reflecting growing clinical screening for asthma and food allergies. Growing incorporation of allergy testing into occupational health screening programs in industries such as agriculture, food processing, and chemical manufacturing drives the demand for diagnostics and therapeutics.
Type Insights
The diagnostics segment is estimated to grow at a CAGR of 10.37% during the forecast period. Growth is supported by increasing adoption of component-resolved diagnostics (CRD), which enables precise identification of individual allergen molecules and improves differentiation between true allergies and cross-reactivity. Expanding use of multiplex microarray testing platforms allows simultaneous detection of multiple allergen sensitivities, improving diagnostic efficiency in specialized allergy laboratories.
The therapeutics segment is projected to register a CAGR of 10.13% in the allergy diagnostics and therapeutics market. The segment is expanding due to the growing adoption of antihistamines, mast cell stabilizers, and immunomodulators. These therapies offer improved symptom control for severe allergic conditions such as asthma and chronic urticaria while reducing reliance on long-term corticosteroid treatments.
Allergen Type Insights
The inhaled segment dominated the market with a revenue share of 48.50% in 2025 due to increasing use of targeted inhaled corticosteroid–long-acting bronchodilator combination formulations that deliver localized anti-inflammatory action directly to the respiratory tract. The introduction of smart inhaler devices with digital dose tracking and improved particle-delivery technologies is enhancing treatment adherence and therapeutic efficiency in allergic respiratory conditions.
The drug allergen segment is projected to grow at a CAGR of 10.94% during the forecast period due to increasing clinical adoption of in vitro drug hypersensitivity testing, including specific IgE assays and basophil activation tests, which improve detection of allergic reactions to antibiotics and anesthetics. Rising hospital pharmacovigilance programs are encouraging systematic drug allergy screening before prescribing high-risk medications.
Test Type Insights
The in vivo tests segment is projected to grow at a CAGR of 10.42% during the forecast period due to growing clinical dependence on skin prick and intradermal testing for real-time allergen response evaluation, which helps physicians confirm clinically relevant sensitivities. Improved standardized allergen extracts and automated skin testing devices are further enhancing test accuracy, reproducibility, and efficiency in specialized allergy clinics.
The in vitro tests segment is projected to grow at a CAGR of 11.03% during the forecast period, driven by the adoption of high-sensitivity chemiluminescent immunoassay systems that enable precise detection of low-level allergen-specific antibodies. Expanding use of automated laboratory analyzers integrated with laboratory information systems is also improving testing throughput and diagnostic consistency in large clinical laboratories.
Competitive Landscape
The allergy diagnostics and therapeutics market is fragmented, with numerous global pharmaceutical companies, diagnostic technology providers, allergen immunotherapy manufacturers, and regional laboratory service providers competing across the value chain. Major companies such as Thermo Fisher Scientific, Siemens Healthineers, Danaher Corporation, Sanofi, and AbbVie maintain strong positions through advanced diagnostic platforms and biologic therapies. The specialized immunotherapy providers and emerging biotechnology firms focus on allergen vaccines, molecular diagnostics, and precision allergy treatments. Competition is driven by technological innovation and increasing investment in targeted biologics and next-generation immunotherapy solutions.
List of Key and Emerging Players in Allergy Diagnostics and Therapeutics Market
- Thermo Fisher Scientific, Inc.
- DASIT Group SPA
- AESKU.GROUP GmbH
- bioMérieux
- Lilly
- HYCOR Biomedical
- Minaris Medical America, Inc.
- Omega Diagnostics Group PLC
- Danaher
- Alcon
- AbbVie, Inc.
- Sanofi
- Allergy Therapeutics
- Pfizer, Inc.
- GSK plc
- Sun Pharmaceutical Industries Ltd
- Hoffmann-La Roche Ltd.
Latest News on Key and Emerging Players
| TIMELINE | COMPANY | DEVELOPMENT |
|---|---|---|
|
January 2026 |
GSK |
GSK announced a USD 2.2 billion acquisition of RAPT Therapeutics, a biotechnology company developing treatments for food allergies. The deal permits GSK access to ozureprubart, an anti-IgE monoclonal antibody currently in Phase II clinical trials for allergies to peanuts, milk, eggs, and other foods. |
|
December 2025 |
DBV Technologies |
DBV Technologies reported positive Phase 3 results for its Viaskin Peanut immunotherapy patch targeting peanut allergies in children aged 4–7. In the trial, 46.6% of treated patients showed improved tolerance to peanut protein compared with 14.8% in the placebo group. |
Source: Secondary Research
Report Scope
| Report Metric | Details |
|---|---|
| Market Size in 2025 | USD 37.03 billion |
| Market Size in 2026 | USD 40.60 billion |
| Market Size in 2034 | USD 85.94 billion |
| CAGR | 9.83% (2026-2034) |
| Base Year for Estimation | 2025 |
| Historical Data | 2022-2024 |
| Forecast Period | 2026-2034 |
| Report Coverage | Revenue Forecast, Competitive Landscape, Growth Factors, Environment & Regulatory Landscape and Trends |
| Segments Covered | By Type, By Allergen Type, By Test Type |
| Geographies Covered | North America, Europe, APAC, Middle East and Africa, LATAM |
| Countries Covered | US, Canada, UK, Germany, France, Spain, Italy, Russia, Nordic, Benelux, China, Korea, Japan, India, Australia, Taiwan, South East Asia, UAE, Turkey, Saudi Arabia, South Africa, Egypt, Nigeria, Brazil, Mexico, Argentina, Chile, Colombia |
Download Free Sample Report to Get Detailed Insights.
Allergy Diagnostics and Therapeutics Market Segments
By Type
-
Diagnostics
- Instruments
- Consumables
- Services
-
Therapeutics
- Antihistamines
- Decongestants
- Corticosteroids
- Mast Cell Stabilizers
- Leukotriene Inhibitors
- Nasal Anti-cholinergic
- Immuno-modulators
- Epinephrine
- Other Therapeutics
By Allergen Type
- Food
- Inhaled
- Drug
- Other Allergen Types
By Test Type
-
In Vivo Tests
- Skin Prick Tests
- Intradermal Tests
- Patch Tests
- In Vitro Tests
By Region
- North America
- Europe
- APAC
- Middle East and Africa
- LATAM
Frequently Asked Questions (FAQs)
Author's Details
Debashree B
Healthcare Lead
Debashree Bora is a Healthcare Lead with over 7 years of industry experience, specializing in Healthcare IT. She provides comprehensive market insights on digital health, electronic medical records, telehealth, and healthcare analytics. Debashree’s research supports organizations in adopting technology-driven healthcare solutions, improving patient care, and achieving operational efficiency in a rapidly transforming healthcare ecosystem.










