Connected Medical Device Market Size, Share & Trends Analysis Report Product Type (Wearable Devices, Implantable Devices, Stationary Devices, Patient Monitoring Devices, Others), By Application (Diagnostics, Remote Patient Monitoring and Telehealth, Treatment Services, Wellness Services, Fitness, Others), By End-user (Hospitals and Clinics, Ambulatory Centers, Nursing Homes, Home Care, Others) and By Region (North America, Europe, APAC, Middle East and Africa, LATAM) Forecasts, 2026-2034
Connected Medical Device Market Overview
The global connected medical device market size is estimated at USD 72.49 billion in 2025, and is projected to reach USD 255.57 billion by 2034 at a CAGR of 15.07% during the forecast period. Strong growth of the market is due to the rising consumer demand for real time health monitoring solutions. Abbott’s Q2 2025 performance exemplifies this trend, with sales of its continuous glucose monitoring (CGM) devices, including FreeStyle Libre and Lingo, surging by 21.4% to USD 1.9 billion, reflecting strong adoption of personalized health data tools.
Key Market Trends & Insights
- North America held a dominant share of the global market with a market share of 38.22% in 2025.
- The Asia Pacific region is growing at the fastest pace, with a CAGR of 17.01%.
- Based on product, the wearable devices segment led, holding a market share of 49.15% in 2025.
- Based on application, the remote patient monitoring and telehealth segment is estimated to grow at a CAGR of 15.66%.
- Based on end user, the hospitals segment led, holding a market share of 45.08% in 2025.
- The U.S. dominates the medical implants market, valued at USD 22.20 billion in 2024 and reaching USD 25.45 billion in 2025.
Table: U.S. Connected Medical Device Market Size (USD Million)
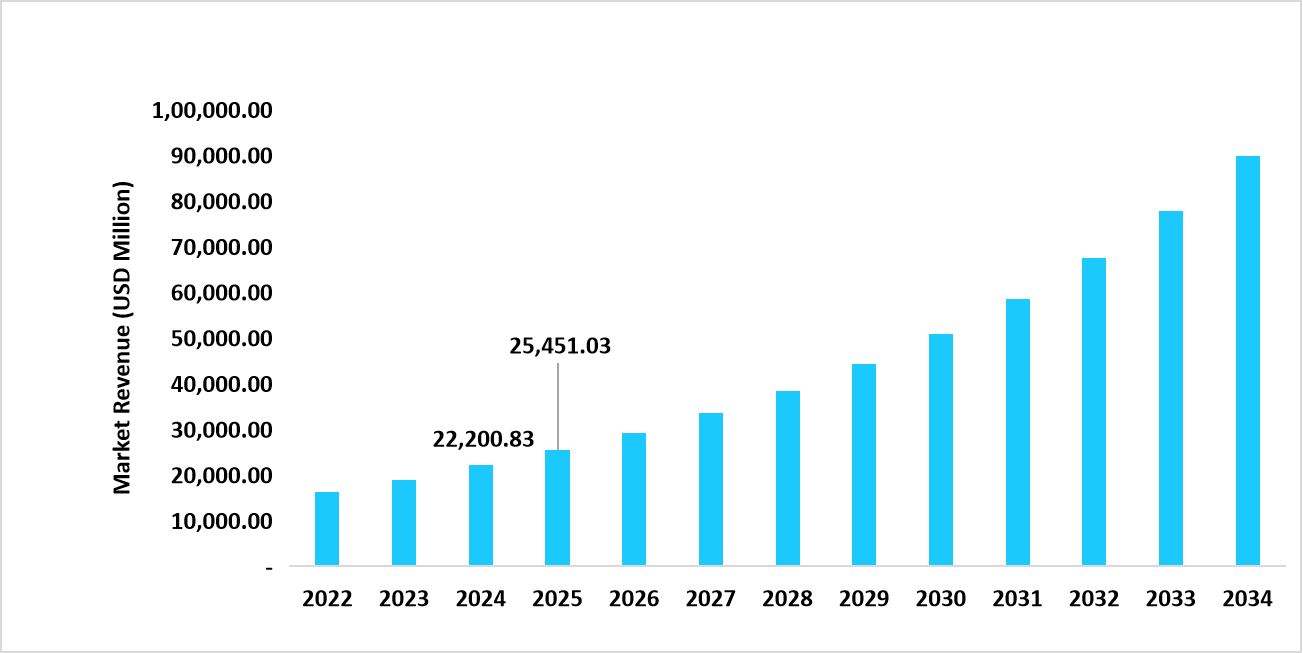
Source: Straits Research
Market Size & Forecast
- 2025 Market Size: USD 110.53 billion
- 2034 Projected Market Size: USD 196.94 billion
- CAGR (2026-2034): 6.67%
- North America: Largest market in 2025
- Fastest Growing Region: Asia Pacific
The global connected medical devices market comprises a wide array of technologies designed to monitor, diagnose, and manage patient health through real-time data exchange, thereby enhancing clinical outcomes and enabling personalized healthcare delivery. By product type, the market includes wearable, implantable, stationary, and patient monitoring devices, with smartwatches and activity trackers dominating due to rising health awareness and integration of advanced biosensors, while wearable patches are gaining traction for continuous glucose and temperature monitoring. Implantable devices such as cardiovascular, orthopedic, and neurological implants are expanding rapidly with growing incidences of chronic diseases and advancements in wireless telemetry and biosensing, whereas stationary devices like infusion pumps, ventilators, and dialysis machines are increasingly connected to improve safety and remote operability. Patient monitoring devices, including vital signs, neurological, and respiratory monitors, are witnessing surging adoption amid the rise of home-based and continuous care models. By application, diagnostics, remote patient monitoring and telehealth, treatment, wellness, and fitness services form the core segments, with remote monitoring and telehealth leading growth due to the increasing demand for virtual healthcare and early disease detection. Among end users, hospitals and clinics hold the largest share driven by advanced infrastructure and high patient inflow, while ambulatory centers and home care are emerging rapidly with the shift toward decentralized and cost-effective healthcare delivery. Overall, continuous advancements in IoT integration, AI-driven analytics, cybersecurity, and wearable and implantable technologies are propelling the robust expansion of the global connected medical devices market.
Latest Market Trends
Integration of AI and Cloud Analytics in Medical Devices
The integration of AI and cloud analytics in medical devices is transforming healthcare by enabling real-time monitoring, predictive diagnostics, and personalized patient care, drives the market growth. Recently, ASUS introduced HealthAI Genie, a generative AI health assistant integrated with the VivoWatch smartwatch. It analyzed real time data to provide personalized insights, tailored recommendations, and proactive monitoring, enhancing patient engagement and enabling efficient healthcare delivery across clinical and home settings. This innovation highlighted the growing potential of AI driven connected devices to improve outcomes and advance preventive care.
Advancements in IoT-Enabled Medical Devices
The adoption of IoT enabled medical devices is transforming healthcare by enabling real time patient monitoring, remote diagnostics, and seamless data sharing across healthcare ecosystems. Recently, Camgenium launched its Soft Silicon platform, enabling rapid, secure, and compliant connected medical device innovation. This platform delivered advanced firmware, cloud integration, and lifecycle management for Nordic Semiconductor devices, facilitating the development of medical IoT solutions. Such innovations enhanced operational efficiency, patient outcomes, and predictive healthcare capabilities globally.
Market Summary
| Market Metric | Details & Data (2025-2034) |
|---|---|
| 2025 Market Valuation | USD 72.49 Billion |
| Estimated 2026 Value | USD 83.15 Billion |
| Projected 2034 Value | USD 255.57 Billion |
| CAGR (2026-2034) | 15.07% |
| Study Period | 2022-2034 |
| Dominant Region | North America |
| Fastest Growing Region | Asia Pacific |
| Key Market Players | Medtronic, Abbott, Koninklijke Philips N.V.,, Siemens Healthineers AG, Boston Scientific Corporation |

Download Free Sample Report to Get Detailed Insights.
Market Drivers
Regulatory Approvals
Regulatory approvals are accelerating market growth by ensuring faster access to innovative devices while maintaining patient safety. Recently, EMA introduced a scientific advice pathway for Class III and active Class IIb devices, offering early guidance on clinical studies under MDR Article 61(2) to enhance innovation and compliance. Such initiatives streamlined approval, boost industry confidence, and encouraged investment.
Market Restraints
Data security and Privacy Concerns
The surge in connected medical devices has increased healthcare data security risks. Recently, 185 million breached records were reported, largely linked to IoT-enabled devices. In August 2025, Modat uncovered a breach affecting 1.2 million devices worldwide, exposing MRI scans and X-rays due to weak safeguards. Similarly, the Change Healthcare ransomware attack compromised the data of over 100 million individuals. These incidents underscored the pressing need for advanced cybersecurity strategies to protect patient data in interconnected healthcare ecosystems.
Market Opportunities
Expansion of Telemedicine and Remote Patient Monitoring Solutions
The expansion of telemedicine and remote patient monitoring (RPM) presents a market opportunity by improving healthcare access and outcomes, particularly in rural and underserved regions. Recently, Telangana's Sanjeevani telemedicine initiative delivered virtual consultations to over 2.23 crore people, including remote tribal populations, with projections to reach 87 lakh users by March 2025. Such development highlighted the potential for innovation, investment, and adoption of connected healthcare solutions globally.
Regional Analysis
The North American region dominated the market with a revenue share of 38.22% in 2025. The growth is attributed to the advanced healthcare infrastructure, and growing reliance on telehealth and remote monitoring solutions. A notable example is Cardinal Health’s June 2025 launch of the Kendall DL Multi System in the U.S., a single-patient use monitoring cable and lead wire system that continuously tracks cardiac activity, blood oxygen levels, and temperature through one point of connection.
U.S. Market: The U.S. market is growing due to rising demand for remote monitoring and chronic disease management. For instance, Abbott launched the FreeStyle Libre 2 Plus sensor in the US in January 2024, which integrates with Tandem Diabetes Care Inc. t: slim X2 insulin pumps. Coupled with expanding telehealth adoption and strong healthcare infrastructure, such innovations drive widespread acceptance of connected medical devices across the country.
Asia Pacific Market Insights
The Asia Pacific region is the fastest growing region with a CAGR of 17.01% during the forecast timeframe, supported by increasing smartphone penetration and digital infrastructure that are driving the adoption of wearables and remote monitoring solutions across urban and rural areas.
India’s market is expanding rapidly, driven by innovations like Renalyx’s AI-based smart hemodialysis machine, which offers affordable, cloud-based renal care in underserved regions. Simultaneously, the country faces a surging cancer burden, with cases expected to reach 1.57 million by 2025, fueling demand for advanced diagnostics and treatment technologies and strengthening India’s position as a key regional growth hub.
By Region Market Share (in percent share %), 2025
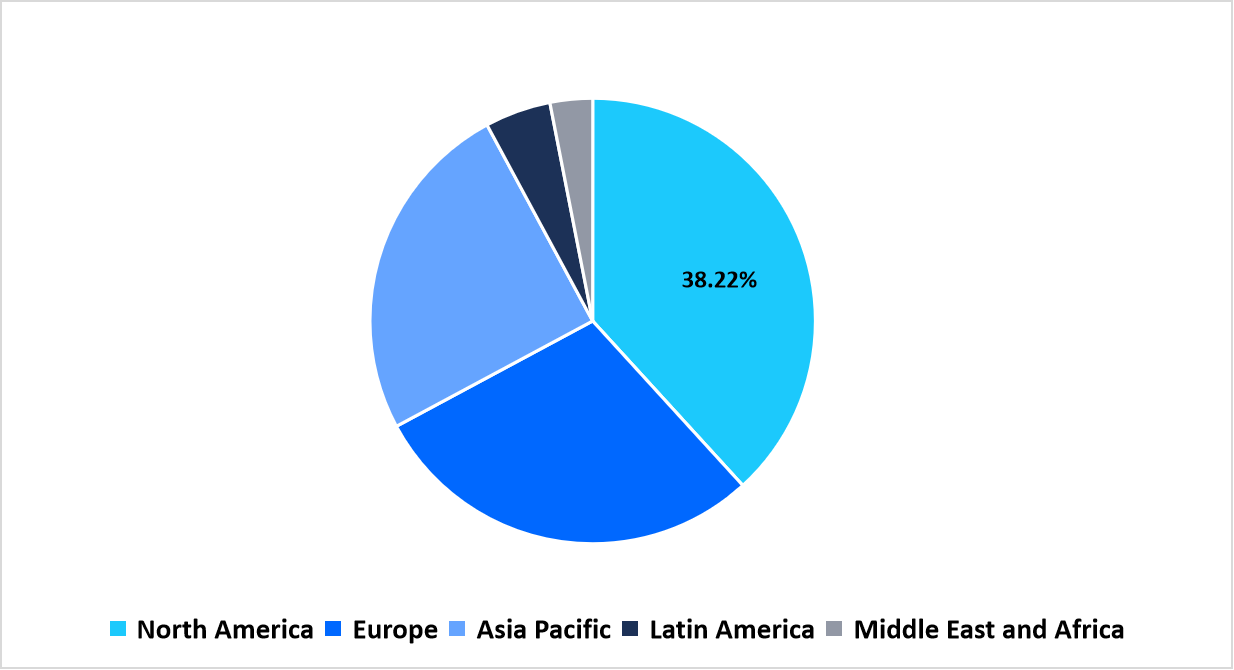
Source: Straits Research
Europe Market Insights
The Europe market is driven by rising adoption of digital health, AI, and IoMT. Regulatory reforms, including the MHRA’s Early Access Service launched in July 2025, allow conditional approvals for innovative devices before full certification. Alongside international reliance routes and safety monitoring, these initiatives enhance NHS adoption, accelerate patient access, and reinforce the Europe’s MedTech innovation leadership, thereby driving the regional market expansion.
Germany's market is growing rapidly, fueled by strong government initiatives like the Digital Healthcare Act and innovative startups. For instance, in February 2025, Smedo introduced contactless vital-sign monitoring technology, enabling real-time tracking of heart rate and respiration. Such breakthroughs highlight Germany’s role as a hub for emerging MedTech startups, fostering advanced remote patient monitoring and digital healthcare adoption.
Middle East and Africa Market Insights
The Middle East and Africa connected medical devices market is being uniquely driven by the rapid emergence of smart hospital ecosystems, fueled by public private partnerships (PPPs) and government backed digital health programs. Countries such as the UAE, Saudi Arabia, and South Africa are investing heavily in national e-health infrastructures that mandate the integration of connected devices into hospital information systems.
Government supported programs, such as the National Digital Health Strategy (2024–2029), encouraged the use of Bluetooth and cloud enabled medical devices to link patients with clinicians through telemedicine hubs, which further enhances the market growth. This creates demand for affordable, interoperable connected devices capable of transmitting real-time health data over mobile networks.
Latin America Market Insights
In Latin America, the connected medical devices market is being uniquely propelled by the growing participation of private health insurers and managed care organizations in remote patient monitoring (RPM) programs. With rising healthcare costs and hospital overcrowding, insurers across countries like Brazil, Mexico, and Chile are incentivizing the use of connected wearables, glucose monitors, and cardiac sensors to reduce readmissions and manage chronic diseases outside clinical settings.
In Argentina, a key driver for the connected medical devices market is the government led digital transformation of elderly care and chronic disease monitoring under the Plan Nacional de Telesalud and regional smart health initiatives.
Product Insights
The wearable devices segment dominated the market with a revenue share of 49.15% in 2025. This segment's prominence is fueled by the widespread use of smart watches, activity trackers, and patches that provide real-time health insights, enhance patient engagement, and support preventive care through continuous monitoring.
The patient monitoring devices segment is anticipated to register the fastest CAGR of 16.07% during the forecast period, due to the rising adoption of remote and continuous monitoring systems, increasing prevalence of chronic diseases, and the growing shift toward telehealth and home-based care models.
Application Insights
The remote patient monitoring and telehealth segment is expected to grow at a CAGR of 15.66%. This market growth is driven by rising chronic disease cases, demand for virtual consultations, and the need for seamless real-time data sharing, improving healthcare accessibility, efficiency, and personalized treatment delivery.
The diagnostics segment dominated the market in 2025, with a revenue share of 27.28%, owing to the rising adoption of connected diagnostic systems for early disease detection, real-time data sharing, and improved clinical accuracy. The increasing use of IoT enabled diagnostic devices, such as connected ECGs, glucose monitors, and imaging systems, has enabled faster diagnosis and seamless integration with electronic health records (EHRs), enhancing workflow efficiency in hospitals and clinics.
End user Insights
The hospitals and clinics segment dominated the market with a revenue share of 45.08% in 2025. This dominance is supported by advanced medical infrastructure, integration of connected devices, and emphasis on precise diagnosis, effective treatment, and coordinated patient care to enhance outcomes and optimize healthcare services.
The home care segment is anticipated to register the fastest CAGR of 16.12% during the forecast period, due to the rising adoption of remote patient monitoring (RPM), increasing prevalence of chronic illnesses, and the growing preference for personalized, cost-effective healthcare outside traditional hospital settings.
By End User Market Share (in percent share %), 2025
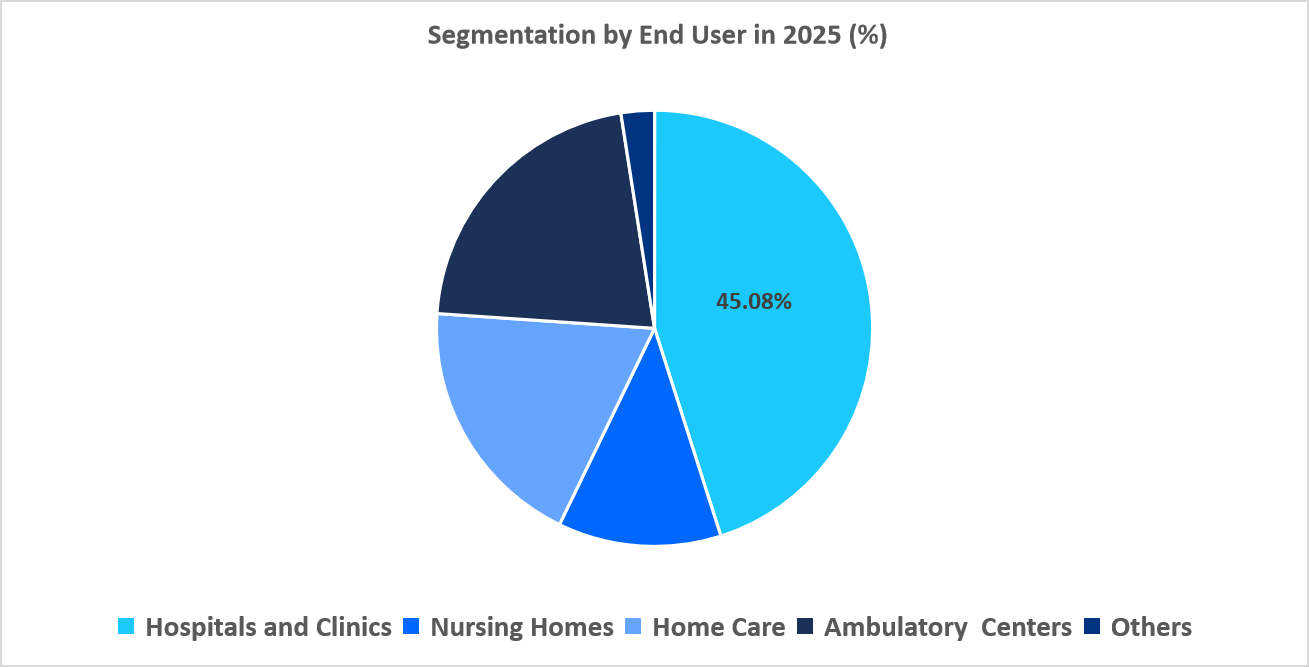
Source: Straits Research
Competitive Landscape
The global connected medical device market is highly fragmented in nature due to numerous global and regional players, rapid technological innovations, diverse product offerings, and varying regulatory standards across different healthcare systems.
Hilo (formerly Aktiia)
Founded in Switzerland in 2018 as Aktiia and rebranded to Hilo in 2025, the company develops AI-powered, cuffless blood-pressure monitoring wearables using optical sensors. It has sold 120,000 + units and raised over USD 100 million.
- In July 2025, Hilo (formerly Aktiia) secured FDA clearance for its Hilo Band, the first cuffless blood pressure monitoring wearable authorized for over-the-counter (OTC) sale in the U.S. The device leveraged optical sensors and AI to analyze arterial pulses, enabling continuous blood pressure tracking without a cuff. Already CE-certified and available in Europe, the Hilo Band is set to launch for U.S. consumers in 2026.
List of Key and Emerging Players in Connected Medical Device Market
- Medtronic
- Abbott
- Koninklijke Philips N.V.,
- Siemens Healthineers AG
- Boston Scientific Corporation
- GE Healthcare
- Hoffmann-La Roche Ltd
- Dexcom, Inc.
- Fitbit
- Garmin Ltd.
- Omron Healthcare, Inc.
- Stryker
- Honeywell International Inc.
- BD
- IMD - International Institute for Management Development
- iRhythm Inc.
- AliveCor, Inc.
- Masimo
- BioTelemetry, a Philips Company
- NIHON KOHDEN CORPORATION
- Others
Strategic Initiatives
- August 2025- Samsung, via its subsidiary NeuroLogica, launched a new range of AI-powered mobile CT systems in India, including CereTom Elite, OmniTom Elite, and BodyTom 32/64. These portable systems enabled advanced diagnostic imaging in ICUs, operating rooms, and emergency departments, improving accessibility in underserved areas.
- May 2025- Pearl Inc. obtained FDA clearance for its AI enabled Second Opinion BLE device. The system analyzed medical images to support radiologists, enhancing diagnostic accuracy and improving clinical decision-making in healthcare settings.
- April 2025- DexCom received FDA clearance for its new G7 15-Day continuous glucose monitoring (CGM) system, designed for adults 18+. The device allows continuous glucose tracking for up to 15.5 days, improving convenience and patient compliance. It is expected to launch in the U.S. in H2 2025.
Report Scope
| Report Metric | Details |
|---|---|
| Market Size in 2025 | USD 72.49 Billion |
| Market Size in 2026 | USD 83.15 Billion |
| Market Size in 2034 | USD 255.57 Billion |
| CAGR | 15.07% (2026-2034) |
| Base Year for Estimation | 2025 |
| Historical Data | 2022-2024 |
| Forecast Period | 2026-2034 |
| Report Coverage | Revenue Forecast, Competitive Landscape, Growth Factors, Environment & Regulatory Landscape and Trends |
| Segments Covered | Product Type, By Application, By End-user |
| Geographies Covered | North America, Europe, APAC, Middle East and Africa, LATAM |
| Countries Covered | US, Canada, UK, Germany, France, Spain, Italy, Russia, Nordic, Benelux, China, Korea, Japan, India, Australia, Taiwan, South East Asia, UAE, Turkey, Saudi Arabia, South Africa, Egypt, Nigeria, Brazil, Mexico, Argentina, Chile, Colombia |
Download Free Sample Report to Get Detailed Insights.
Connected Medical Device Market Segments
Product Type
-
Wearable Devices
- Activity Trackers
- Smart Watches
- Patches
- Others
-
Implantable Devices
- Cardiovascular Implants
- Orthopedic Implants
- Neurological Implants
- Others
-
Stationary Devices
- Infusion Pumps
- Ventilators
- Dialysis Machines
- Others
-
Patient Monitoring Devices
- Vital Signs Monitors
- Neurological Monitors
- Respiratory Monitors
- Others
- Others
By Application
- Diagnostics
- Remote Patient Monitoring and Telehealth
- Treatment Services
- Wellness Services
- Fitness
- Others
By End-user
- Hospitals and Clinics
- Ambulatory Centers
- Nursing Homes
- Home Care
- Others
By Region
- North America
- Europe
- APAC
- Middle East and Africa
- LATAM
Frequently Asked Questions (FAQs)
Author's Details
Debashree B
Healthcare Lead
Debashree Bora is a Healthcare Lead with over 7 years of industry experience, specializing in Healthcare IT. She provides comprehensive market insights on digital health, electronic medical records, telehealth, and healthcare analytics. Debashree’s research supports organizations in adopting technology-driven healthcare solutions, improving patient care, and achieving operational efficiency in a rapidly transforming healthcare ecosystem.










