Contrast Media Injectors Market Size, Share & Trends Analysis Report By Product (Injector Systems, Consumables, Tubing, Syringe, Others), By Type (Single-head Injectors, Dual-head Injectors, Syringeless Injectors), By Application (Radiology, Interventional Cardiology, Interventional Radiology), By End Use (Hospitals, Diagnostic Centers, Ambulatory Surgery Centers) and By Region (North America, Europe, APAC, Middle East and Africa, LATAM) Forecasts, 2026-2034
What is the Size of Contrast Media Injectors Market?
The contrast media injectors market size was valued at USD 2.45 billion in 2025 and is projected to grow from USD 2.68 billion in 2026 to USD 5.74 billion by 2034, growing at a CAGR of 9.95% during the forecast period.
Key Market Insights
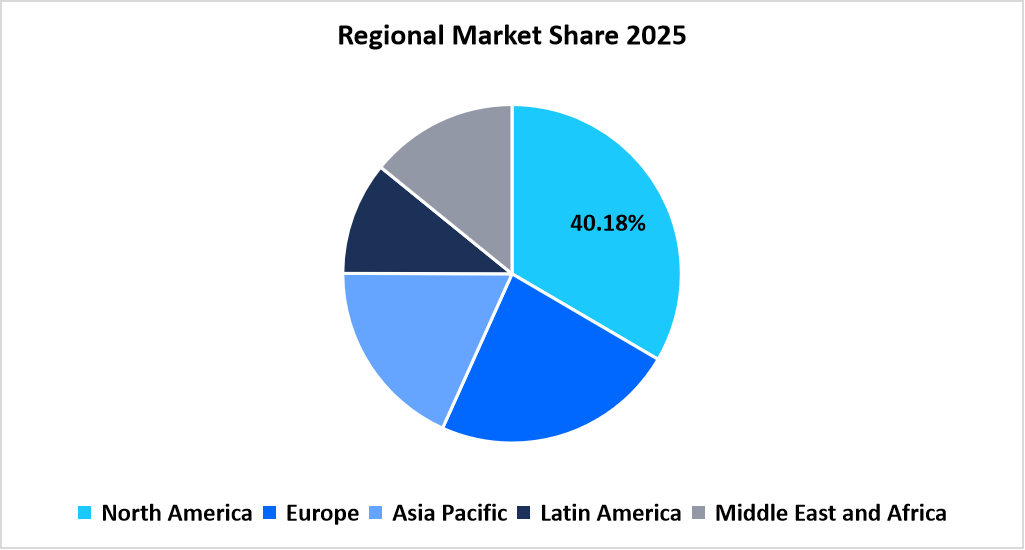
- North America dominated the contrast media injectors market with the largest share of 40.18% in 2025.
- The Asia Pacific is expected to be the fastest-growing region in the contrast media injectors market during the forecast period at a CAGR of 11.95%.
- By product, the injector systems segment is expected to register a CAGR of 10.45% during the forecast period.
- By type, the single-head injectors segment dominated the market with a revenue share of 40.13% in 2025.
- By application, the radiology segment dominated the market with a revenue share of 35.67% in 2025.
- By end use, the hospitals segment dominated the market with a revenue share of 45.44% in 2025.
- The US contrast media injectors market size was valued at USD 860.73 million in 2025 and is projected to reach USD 943.27 million in 2026.
Market Summary
| Market Metric | Details & Data (2025-2034) |
|---|---|
| 2025 Market Valuation | USD 2.45 billion |
| Estimated 2026 Value | USD 2.68 billion |
| Projected 2034 Value | USD 5.74 billion |
| CAGR (2026-2034) | 9.95% |
| Study Period | 2022-2034 |
| Dominant Region | North America |
| Fastest Growing Region | Asia Pacific |
| Key Market Players | Shenzhen Boon Medical Supply Co., Ltd., Bayer, Bracco Diagnostics Inc., Ulrich GmbH & Co. KG, Guerbet LLC |

Download Free Sample Report to Get Detailed Insights.
What are the Current Trends in Contrast Media Injectors Market?
Growing shift toward dual-head and multi-head injector systems that allow precise sequencing of contrast media and saline flush, improving bolus control and image quality in advanced CT applications. There is increasing integration of injectors with imaging modalities such as CT and MRI, enabling automated bolus tracking, protocol synchronization, and more consistent scan timing across patients.
Increasing focus on smart injector technologies with real-time pressure monitoring, extravasation detection, and automated dose calculation enhance patient safety and reduce contrast-related complications. The market is also seeing wider adoption of MRI-compatible and MRI-conditional injector systems, driven by rising use of high-field MRI scanners and contrast-enhanced MRI procedures.
Rising interventional radiology and cardiology procedures is supporting demand for high-pressure angiographic injectors that can operate in hybrid operating rooms and catheterization labs. At the same time, increasing global volumes of cardiovascular and oncology imaging are pushing healthcare providers to invest in high-throughput, workflow-efficient injector platforms.
Increasing emphasis on dose optimization and contrast utilization efficiency leads to hospitals seeking affordable solutions that comply with safety guidelines. Vendors are introducing user-friendly interfaces, disposable syringe systems, and connectivity features that support standardization, reduce setup time, and improve overall imaging workflow efficiency.
What are the Driving Factors of Contrast Media Injectors Market?
Increasing hospital investments in advanced imaging infrastructure strengthens procurement of high-performance injector systems to support growing diagnostic workloads.
Rising healthcare spending in emerging economies expands access to advanced diagnostic imaging and creates increasing supply activity for injector manufacturers.
Expanding interventional radiology and catheterization laboratories increases requirements for high-pressure injectors and supports equipment upgrades in hybrid operating rooms.
Growing adoption of protocol standardization in radiology departments raises the need for automated systems that deliver consistent contrast volumes and flow rates across high patient volumes.
Which Factors are Hampering the Growth of Contrast Media Injectors Market?
High upfront capital investment and recurring disposable costs restrain the market, as purchasing decisions remain closely linked to hospital imaging equipment capital cycles. This results in delayed procurement and slower adoption of contrast media injector systems under constrained hospital budgets.
Clinical risks associated with contrast-induced nephropathy and hypersensitivity reactions limit market growth. This reduces the use of contrast-enhanced imaging in high-risk patient populations, leading to lower injector utilization rates.
Stringent regulatory and compliance requirements hinder market expansion, as injector systems classified as Class II or Class III devices by the US Food and Drug Administration and subject to ISO and European MDR standards face lengthy approval timelines and post-market surveillance obligations. This increases manufacturer costs and slows product commercialization.
What are the Growth Opportunities in Contrast Media Injectors Market?
Advancements in AI-based lesion detection and automated image reconstruction create new opportunities, as injector systems integrated with AI-driven bolus timing algorithms improve diagnostic accuracy and workflow precision. Investments in AI-enabled imaging ecosystems open integration opportunities for smart injector platforms.
Expansion of freestanding diagnostic imaging centers opens new market opportunities, as rising outpatient imaging volumes increase demand for compact, cost-efficient, and easy-to-operate injector systems, supporting targeted product positioning toward standalone radiology clinics.
The growth of minimally invasive image-guided therapies creates long-term opportunities, as increasing adoption of procedures such as transcatheter aortic valve replacement and neurointerventional interventions drives demand for high-pressure angiography injectors.
Regional Analysis
North America Contrast Media Injectors Market
The contrast media injectors market in North America held the largest share of 40.18% in the market in 2025 due to high diagnostic imaging utilization and early adoption of workflow-standardized radiology infrastructure. Contrast-enhanced CT and MRI scans remain among the most frequently reimbursed imaging procedures under Medicare Part B. Regulatory clearance data released by the US Food and Drug Administration confirm sustained approvals for contrast injector systems across multiple imaging modalities.
Asia Pacific Contrast Media Injectors Market
Asia Pacific represents the fastest expanding region for contrast media injectors, with a CAGR of 11.95%. This growth is supported by rapid diagnostic imaging capacity growth and public healthcare capital expenditure. According to the World Health Organization, countries across the Western Pacific region continue to scale advanced imaging access to address rising noncommunicable disease burden, particularly cancer and cardiovascular disorders. Government programs focus on hospital infrastructure expansion initiatives and deployment of high CT density per million population. Sustained imaging investments accelerate demand for automated contrast injector systems across tertiary and secondary care facilities.
Source: Straits Analysis
Europe Contrast Media Injectors Market
The market in Europe is driven by the high diagnostic imaging volumes supported by the statutory health insurance system, which covers nearly 90% of the population, ensuring broad and reimbursed access to CT and MRI procedures. Germany records among the highest numbers of MRI examinations in Europe and maintains one of the highest scanner densities per million population, reflecting substantial imaging throughput that directly drives demand for automated contrast injector systems.
Middle East & Africa Contrast Media Injectors Market
The Middle East & Africa market is growing due to the adoption of advanced imaging modalities and the expansion of diagnostic imaging infrastructure. Under Vision 2030, the UAE is investing heavily in new hospitals, medical cities, and specialized diagnostic centers, accelerating installations of high-slice CT scanners, MRI systems, and hybrid imaging platforms across tertiary care facilities. As imaging capacity expands, healthcare providers are increasingly adopting automated, dual-head, and syringeless contrast injector technologies to ensure accurate dosing, reduced contrast wastage, and seamless integration with radiology IT systems.
Latin America Contrast Media Injectors Market
The market in Latin America is growing due to increasing private healthcare spending and the expansion of diagnostic centers, alongside continuous technological advancements in contrast injector systems. Public healthcare system expansion across major LATAM countries supports imaging demand, as governments in countries such as Brazil and Mexico continue to scale universal healthcare coverage, increasing patient access to CT, MRI, and angiography services that require contrast injectors. Hospital infrastructure modernization programs increase equipment replacement rates, as aging imaging suites across public and private hospitals undergo phased upgrades, creating steady demand for newer-generation injector systems compatible with modern scanners.
Product Insights
The injector systems segment is expected to register a CAGR of 10.45% during the forecast period due to the increasing demand for advanced imaging procedures such as CT, MRI, and cardiovascular angiography, which hence is driving adoption of the injector systems. Technological advancements, including automated dosing, enhanced safety mechanisms, and improved workflow efficiency, are further supporting growth in this segment.
The consumables segment is expected to register a CAGR of 10.56% during the forecast period. Continuous and repeated use of tubing, syringes, and related components in diagnostic imaging procedures generates recurring demand, which hence accelerates the market growth. Rising procedure volumes and strict infection control protocols are key factors enhancing the expansion of consumables segment.
Type Insights
Single-head injectors dominated the type segment with a revenue share of 40.13% in 2025. Their widespread use in routine diagnostic imaging, cost-effectiveness, and ease of operation contribute to their strong market position. These systems are commonly preferred in standard CT and MRI procedures, particularly in facilities with moderate imaging volumes, which further supports market growth.
The syringeless injectors segment is expected to register a notable growth with a CAGR of 10.66% during the forecast period. The growth is attributed to several advantages such as reduced contrast media wastage, enhanced workflow efficiency, and lower risk of contamination, which are driving increased adoption of syringeless injectors.
Application Insights
Radiology held the largest share of the application segment, accounting for 35.67% in 2025. The high volume of diagnostic imaging procedures, including CT and MRI scans, contributes to the dominance of the radiology segment. Increasing prevalence of chronic diseases and the growing need for early and accurate diagnosis are further boosting demand in radiology applications.
The interventional cardiology segment is anticipated to grow at a CAGR of 10.81% during the forecast period. The growth is attributed to the rising incidence of cardiovascular diseases and increasing adoption of minimally invasive cardiac procedures. In addition, contrast media injectors play a critical role in angiography and catheter-based interventions, supporting segment growth.
End Use Insights
Hospitals dominated the market with a share of 45.44% in 2025 due to the availability of advanced imaging infrastructure, high patient inflow, and skilled healthcare professionals. Hospitals also conduct a wide range of diagnostic and interventional procedures requiring contrast media injectors, which further fuels segment growth.
The diagnostic centers segment is projected to grow at a CAGR of 10.33% during the forecast period. Increasing preference for outpatient imaging services, expanding private diagnostic networks, and shorter wait times are driving the growth of diagnostic centers.
Competitive Landscape
The global contrast media injectors market is moderately concentrated, characterized by the presence of multinational medical device manufacturers along with a growing base of regional equipment suppliers. Competition in the market is shaped by injector performance parameters such as flow rate precision, pressure control, and compatibility with CT, MRI, and angiography systems. Product portfolios covering single- and dual-head injectors, scalability across diagnostic imaging modalities, and alignment with hospital workflow requirements influence market dynamics. Leading companies sustain their positions through installed base expansion in hospitals and diagnostic centers, long-term service agreements, system-level integration with imaging platforms, and adherence to regulatory and safety standards across major healthcare markets.
List of Key and Emerging Players in Contrast Media Injectors Market
- Shenzhen Boon Medical Supply Co., Ltd.
- Bayer
- Bracco Diagnostics Inc.
- Ulrich GmbH & Co. KG
- Guerbet LLC
- Braun SE
- Shenzhen Antmed Co., Ltd.
- Ecomed Solutions
- Siemens Medical Solutions USA, Inc.
- GE HealthCare
- Canon Medical Systems Corporation
- ACIST Medical Systems, Inc.
- MEDTRON AG
- Nemoto Kyorindo Co., Ltd.
Latest News on Key and Emerging Players
| TIMELINE | COMPANY | DEVELOPMENT |
|---|---|---|
|
January 2026 |
Bayer |
Bayer presented new Phase III clinical data for its investigational low-dose MRI contrast agent gadoquatrane and advancements across its radiology portfolio at the European Congress of Radiology (ECR) 2026 in Vienna, Austria. |
|
December 2025 |
Bracco Diagnostics Inc. |
Bracco Diagnostics Inc. announced the FDA clearance for the Max 3 rapid exchange syringeless injector, which improves flexibility across CT and MRI workflows. |
|
December 2025 |
Bayer |
Bayer announced that its MEDRAD Centargo multi-patient CT injector received 510(k) clearance from the US Food and Drug Administration (FDA). |
|
November 2025 |
Bracco Diagnostics Inc. |
Bracco Diagnostics Inc., the US subsidiary of Bracco Imaging S.p.A., announced that the US FDA had expanded the indication for its Bracco-branded Max 3 Rapid Exchange and Syringeless Injector for use in magnetic resonance imaging (MRI) procedures. |
Source: Secondary Research
Report Scope
| Report Metric | Details |
|---|---|
| Market Size in 2025 | USD 2.45 billion |
| Market Size in 2026 | USD 2.68 billion |
| Market Size in 2034 | USD 5.74 billion |
| CAGR | 9.95% (2026-2034) |
| Base Year for Estimation | 2025 |
| Historical Data | 2022-2024 |
| Forecast Period | 2026-2034 |
| Report Coverage | Revenue Forecast, Competitive Landscape, Growth Factors, Environment & Regulatory Landscape and Trends |
| Segments Covered | By Product, By Type, By Application, By End Use |
| Geographies Covered | North America, Europe, APAC, Middle East and Africa, LATAM |
| Countries Covered | US, Canada, UK, Germany, France, Spain, Italy, Russia, Nordic, Benelux, China, Korea, Japan, India, Australia, Taiwan, South East Asia, UAE, Turkey, Saudi Arabia, South Africa, Egypt, Nigeria, Brazil, Mexico, Argentina, Chile, Colombia |
Download Free Sample Report to Get Detailed Insights.
Contrast Media Injectors Market Segments
By Product
-
Injector Systems
- CT Injector Systems
- MRI Injector Systems
- Cardiovascular/Angiography Injector Systems
- Consumables
- Tubing
- Syringe
- Others
By Type
- Single-head Injectors
- Dual-head Injectors
- Syringeless Injectors
By Application
- Radiology
- Interventional Cardiology
- Interventional Radiology
By End Use
- Hospitals
- Diagnostic Centers
- Ambulatory Surgery Centers
By Region
- North America
- Europe
- APAC
- Middle East and Africa
- LATAM
Frequently Asked Questions (FAQs)
Author's Details
Dhanashri B
Senior Research Associate
Dhanashri Bhapakar is a Senior Research Associate with 3+ years of experience in the Biotechnology sector. She focuses on tracking innovation trends, R&D breakthroughs, and market opportunities within biopharmaceuticals and life sciences. Dhanashri’s deep industry knowledge enables her to provide precise, data-backed insights that help companies innovate and compete effectively in global biotech markets.










