eClinical Solutions Market Size, Share & Trends Analysis Report By Product (Electronic Data Capture & Clinical Data Management Systems, Clinical Trial Management Systems, Randomization & Trial Supply Management, Electronic Clinical Outcome Assessment, Clinical Analytics & Data-Integration Platforms, Safety & Pharmacovigilance Solutions, Electronic Trial Master File (eTMF), Other Products), By Delivery Mode (Cloud-based, Web-hosted, On-premise), By Clinical Trial Phase (Phase I, Phase II, Phase III, Phase IV), By End User (Pharmaceutical & Biotechnology Companies, Contract Research Organizations, Medical Device Manufacturers, Academic & Research Institutions, Other End Users) and By Region (North America, Europe, APAC, Middle East and Africa, LATAM) Forecasts, 2026-2034
eClinical Solutions Market Size
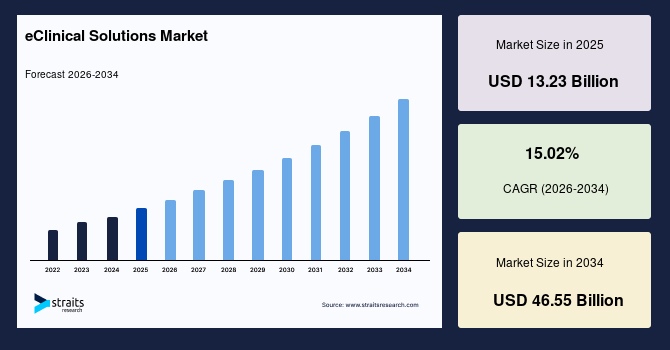
The eClinical solutions market size was valued at USD 13.23 billion in 2025 and is projected to grow from USD 15.20 billion in 2026 to USD 46.55 billion by 2034 at a CAGR of 15.02% during the forecast period (2026-2034),.
The eClinical solutions market is growing steadily due to increasing clinical trial complexity, rising data volumes, and the need for faster drug approvals. According to ClinicalTrials.gov 2025, globally, more than 450,000 clinical studies are registered, highlighting the expanding research landscape. The World Health Organization reports a continuous rise in chronic diseases, which drives higher demand for clinical trials and associated digital tools. Regulatory authorities such as the US Food and Drug Administration promote the use of electronic systems to improve data transparency and compliance. The adoption of decentralized trials and telehealth integration is improving patient participation and retention. Cloud-based platforms, electronic data capture systems, and AI-driven analytics enhance real-time decision-making and reduce operational delays. Integration of wearable devices also supports remote patient monitoring. Thus, digital transformation and patient-centric approaches are key factors driving sustained growth in the market.
Key Market Insights
- North America dominated the eClinical solutions market with the largest share of 47.64% in 2025.
- The Asia Pacific is expected to be the fastest-growing region in the market during the forecast period at a CAGR of 19.04%.
- Based on product, the electronic clinical outcome assessment segment is expected to register a CAGR of 16.78% during the forecast period.
- Based on delivery mode, the cloud-based segment accounted for the 48.13% revenue share in 2025.
- Based on clinical trial phase, the phase I is projected to grow at a CAGR of 16.46% during the forecast period.
- Based on end user, pharmaceutical & biotechnology companies dominate the market, accounting for 57.39% revenue share in 2025.
- The US eClinical solutions market size was valued at USD 5.67 billion in 2025 and is projected to reach USD 6.51 billion in 2026.
Market Summary
| Market Metric | Details & Data (2025-2034) |
|---|---|
| 2025 Market Valuation | USD 13.23 Billion |
| Estimated 2026 Value | USD 15.20 Billion |
| Projected 2034 Value | USD 46.55 Billion |
| CAGR (2026-2034) | 15.02% |
| Study Period | 2022-2034 |
| Dominant Region | North America |
| Fastest Growing Region | Asia Pacific |
| Key Market Players | Oracle, Veeva Systems, IQVIA, Dassault Systèmes, Parexel International Corporation |
Download Free Sample Report to Get Detailed Insights.
Emerging Trends in eClinical Solutions Market
Growing Adoption of Decentralized Clinical Trials
Decentralized clinical trials are becoming a core trend in the eClinical solutions market as they reduce dependency on physical trial sites and improve patient accessibility. These trials use digital platforms, teleconsultations, and home-based diagnostics to collect data remotely. According to ClinicalTrials.gov (2025), a growing share of newly registered studies includes decentralized or hybrid elements. For example, remote oncology trials use wearable sensors and ePRO tools to track patient health continuously, which improves data accuracy and increases patient retention, especially in geographically dispersed populations.
Integration of Real-World Data (RWD) for Enhanced Clinical Decision-making
eClinical platforms increasingly integrate real-world data from electronic health records, insurance claims, and patient registries to improve trial insights. The US Food and Drug Administration highlights the use of RWD to support regulatory decisions and post-market surveillance. For instance, oncology trials now combine clinical data with real-world patient outcomes to better understand long-term drug effectiveness. This approach enhances data accuracy, supports faster approvals, and enables more personalized treatment development in evolving therapeutic areas.
eClinical Solutions Market Drivers
Growing Biopharma R&D Investment and Regulatory Push for Digital Trial Master Files (eTMF) Data Systems Drives Market
Increasing investment in biopharmaceutical research is accelerating demand for advanced digital tools to manage complex clinical development processes. In 2025-2026, major funding activity highlights this shift, with Pfizer raising its R&D spending by around 12% to expand late-stage pipelines, while AstraZeneca committed nearly USD 400 million to new research infrastructure. This surge in funding increases clinical trial volume and data intensity, which drives adoption of electronic data capture, clinical trial management systems, and integrated analytics platforms for efficient, compliant, and faster study execution.
Strict regulatory focus on inspection readiness is driving adoption of eClinical solutions, especially electronic trial master file systems. Authorities like the US Food and Drug Administration and European Medicines Agency require complete, traceable, and audit-ready clinical documentation. Regulatory inspection findings show that incomplete documentation remains a common compliance issue, pushing sponsors toward digital systems. For instance, companies implementing eTMF platforms achieve real-time document tracking, automated audit trails, and standardized workflows, which significantly reduce inspection findings and improve overall regulatory compliance efficiency.
eClinical Solutions Market Restraints
Increasing Cybersecurity Risks, High Implementation Cost, and Limited Reimbursement Restrain eClinical Solutions Market Growth
Rising cybersecurity risks and strict data privacy regulations act as major restraints in the market. Platforms handling sensitive patient data must comply with regulations like those enforced by the GDPR frameworks and other regulations. For instance, clinical trial sponsors in Europe face delays when cloud-based systems require validation for cross-border data transfer compliance. Data breach concerns and high investment in secure infrastructure increase operational costs, making smaller research organizations hesitant to adopt fully digital eClinical systems despite their efficiency benefits.
High implementation costs restrain the market as deploying integrated platforms requires substantial upfront investment in software licensing, cloud infrastructure, data migration, and workforce training. Smaller biotech companies and mid-sized research sites often struggle to afford advanced tools such as electronic data capture and clinical trial management systems. For example, multi-site clinical trials require system customization, integration with existing databases, and ongoing technical support, which significantly increases operational expenses. These cost pressures limit adoption, especially among emerging sponsors, and slow the transition from traditional to fully digital clinical trial environments.
eClinical Solutions Market Opportunities
Expansion of Digital Biomarker-based Clinical Trials and Growth of AI-driven Patient Recruitment Offer Growth Opportunities for eClinical Solutions Market
The growing use of digital biomarkers creates a strong opportunity for eClinical solutions by enabling continuous, objective patient monitoring during trials. Wearable devices now capture real-time data such as heart rate, activity, and sleep patterns, improving endpoint accuracy. The US National Institutes of Health highlights increasing adoption of digital endpoints in ongoing studies. For example, cardiovascular trials use smartwatch-based ECG monitoring to detect irregularities remotely, reducing site visits and enhancing data richness, which drives demand for advanced analytics and integrated eClinical platforms.
AI-based tools are transforming how clinical trial participants and sites are identified, creating a strong opportunity for eClinical platforms. These systems use machine learning to analyze electronic health records, genomic datasets, and historical trial data to quickly match eligible patients and high-performing sites. For example, IQVIA uses its AI-powered recruitment platform to scan large healthcare datasets and identify suitable trial participants faster, while Oracle integrates predictive analytics in its clinical solutions to optimize site selection, reducing enrollment timelines and improving overall study efficiency.
Regional Analysis
North America: Market Leadership through Strong Adoption of Decentralized Trials and Strong Presence of Leading CROs
North America accounted for the largest regional share in the global market, accounting for a share of 47.64% in 2025. This dominance can be attributed to strong adoption of decentralized trial frameworks supported by advanced home-based care infrastructure, high penetration of integrated EHR-clinical trial platforms across large hospital networks, and early use of AI-driven protocol optimization tools by US based CROs. The widespread use of real-world data from insurance-linked health databases enables faster patient stratification and more efficient clinical trial execution.
The strong presence of leading CROs such as IQVIA and Labcorp Drug Development in the US actively deploys advanced digital trial platforms across large-scale studies. High biopharma R&D intensity, with companies like Pfizer and Moderna investing heavily in complex pipelines, increases demand for integrated data systems. The widespread use of claims-linked health datasets and oncology-specific research networks enables precise patient matching and efficient multi-site trial execution. These factors collectively position the US as a dominant country in the North America eClinical solutions market.
The Canada market is supported by the adoption of unified provincial health data systems, which allow seamless integration of clinical trial data with publicly funded healthcare records, improving patient tracking and data consistency. National research networks like Clinical Trials Ontario streamline multi-site study approvals and digital coordination. Additionally, increasing use of bilingual eClinical platforms ensures regulatory and patient compliance across regions, supporting efficient execution of diverse and large-scale clinical trials.
Asia Pacific: Fastest Growth Driven by Expansion of Regional Clinical Trial Hubs and Expansion of Investigator Site Networks
The Asia Pacific eClinical solutions market is expected to register a CAGR of 19.04% during the forecast period, driven by rapid expansion of region-specific clinical trial hubs in countries like China and India, where large treatment-naïve patient pools enable faster enrollment. Government-backed digital health ecosystems, such as national health ID programs, support structured patient data integration, while increasing participation of local biotech firms in global trials drives demand for scalable, cloud-based eClinical platforms.
The market in China is expanding due to rapid adoption of hospital-based clinical research platforms integrated with large public hospital networks, enabling centralized patient data access for trials. Strong government-backed initiatives like National Medical Products Administration digital submission systems accelerate electronic data usage in approvals. Increasing use of site management organizations across urban trial centers improves digital trial coordination and drives demand for scalable eClinical platforms tailored to high-volume, multi-site studies.
Rapid expansion of investigator site networks linked to large tertiary hospitals for high patient recruitment through digitally managed trial workflows is a major factor driving the India market. Government-backed digital health infrastructure like Ayushman Bharat Digital Mission supports the creation of unified health IDs, improving structured patient data availability for trials. India’s large and diverse patient pool, including high prevalence of chronic and infectious diseases, enables faster enrollment and supports demand for scalable eClinical platforms in multi-site trials.
By Product
The electronic data capture & clinical data management systems segment accounted for the largest share of the eClinical solutions market, by product, in 2025, accounting for 31.95% of revenue. The growth is driven by metadata-driven study build, enabling rapid protocol amendments, automated discrepancy management, reducing manual queries, and direct integration with eSource systems capturing real-time site data, improving accuracy and submission readiness.
The electronic clinical outcome assessment segment is expected to grow at a CAGR of 16.78% during the forecast period, fueled by the use of bring-your-own-device models that improve patient compliance, real-time symptom tracking in CNS and pain trials, and platforms like Medidata Solutions eCOA enabling multilingual, remote patient-reported outcome capture across geographically diverse study populations.
By Delivery Mode
Cloud accounted for a 48.13% share of the delivery mode segment in 2025 due to advantages such as real-time data access across global trial sites, rapid scalability for multi-country studies, lower upfront IT infrastructure costs, and seamless integration with ePRO, EDC, and analytics platforms. It also enables faster study startup, centralized monitoring, and efficient collaboration among sponsors, CROs, and investigators.
The on-premise segment is expected to grow at a CAGR of 17.21% during the forecast period, fueled by demand for full data sovereignty among large pharma firms, enabling complete internal control over sensitive trial data. It also supports highly customized legacy system integration and offline data processing capabilities in restricted-network research environments, especially in early-phase trials.
By Clinical Trial Phase
Phase III of the clinical trial phase segment accounted for a dominant share of 40.67% in 2025, as these trials involve large, multi-country patient populations requiring high-volume data capture and real-time monitoring. Complex endpoint validation, frequent protocol amendments, and intensive safety reporting drive extensive use of integrated EDC, eCOA, and CTMS platforms to ensure data consistency, scalability, and regulatory submission readiness.
The Phase I segment is expected to grow at a CAGR of 16.46% during the forecast period, owing to increasing use of adaptive trial designs requiring rapid data analysis, rising first-in-human biologics studies demanding intensive pharmacokinetic monitoring, and specialized early-phase units using integrated eSource and real-time safety tracking systems to manage high-frequency data capture efficiently.
By End User
Pharmaceutical & biotechnology companies dominated the end-user segment with a share of 57.39% in 2025, as they conduct large-scale global trials requiring integrated eClinical platforms for protocol standardization across regions. Increasing reliance on precision medicine pipelines drives high use of biomarker-linked data capture systems. Internal sponsor-led virtual trial operations and direct partnerships with CROs enable faster decision-making and centralized regulatory submission management across complex multi-phase drug development programs.
The contract research organizations segment is expected to have a notable growth with a CAGR of 17.80% during the forecast period due to increasing outsourcing of end-to-end clinical operations by sponsors to reduce development timelines, rising adoption of centralized digital trial management platforms enabling multi-sponsor scalability, and expansion of therapeutic-specialized CRO models in oncology and rare disease studies requiring high-frequency data orchestration and global site coordination.
Competitive Landscape
The eClinical solutions market is moderately consolidated, with major global players dominating through comprehensive digital platforms covering data capture, trial management, and analytics. Key companies include Oracle, IQVIA, Medidata Solutions, Parexel, and Veeva Systems. These firms lead due to strong cloud infrastructure, integrated platforms, and global client networks. Smaller providers compete by offering niche, cost-effective, and customizable solutions for mid-sized sponsors. Competition is increasing through AI integration, strategic partnerships, and platform consolidation to support decentralized and data-driven clinical trials.
List of Key and Emerging Players in eClinical Solutions Market
- Oracle
- Veeva Systems
- IQVIA
- Dassault Systèmes
- Parexel International Corporation
- ICON plc
- eClinical Solutions
- Signant Health
- Clario
- Medable
- Datatrak International
- Anju Software
- MaxisIT
- IBM
- RealTime Software Solutions
- CRF Health
- ERT Clinical
- ArisGlobal
- OpenClinica
Recent Developments
- In October 2025, eClinical Solutions entered a strategic partnership with ZS Associates to combine clinical data platforms (elluminate) with advanced analytics and consulting capabilities.
- In September 2025, IQVIA launched Clinical Trial Financial Suite (CTFS), featuring AI-enabled financial management for clinical trials and integration across fragmented clinical systems.
- In August 2025, IQVIA joined Veeva’s CRO Clinical Data Partner and AI partner programs to enable joint clinical trial execution.
Report Scope
| Report Metric | Details |
|---|---|
| Market Size in 2025 | USD 13.23 Billion |
| Market Size in 2026 | USD 15.20 Billion |
| Market Size in 2034 | USD 46.55 Billion |
| CAGR | 15.02% (2026-2034) |
| Base Year for Estimation | 2025 |
| Historical Data | 2022-2024 |
| Forecast Period | 2026-2034 |
| Report Coverage | Revenue Forecast, Competitive Landscape, Growth Factors, Environment & Regulatory Landscape and Trends |
| Segments Covered | By Product, By Delivery Mode, By Clinical Trial Phase, By End User |
| Geographies Covered | North America, Europe, APAC, Middle East and Africa, LATAM |
| Countries Covered | US, Canada, UK, Germany, France, Spain, Italy, Russia, Nordic, Benelux, China, Korea, Japan, India, Australia, Taiwan, South East Asia, UAE, Turkey, Saudi Arabia, South Africa, Egypt, Nigeria, Brazil, Mexico, Argentina, Chile, Colombia |
Download Free Sample Report to Get Detailed Insights.
eClinical Solutions Market Segments
By Product
- Electronic Data Capture & Clinical Data Management Systems
- Clinical Trial Management Systems
- Randomization & Trial Supply Management
- Electronic Clinical Outcome Assessment
- Clinical Analytics & Data-Integration Platforms
- Safety & Pharmacovigilance Solutions
- Electronic Trial Master File (eTMF)
- Other Products
By Delivery Mode
- Cloud-based
- Web-hosted
- On-premise
By Clinical Trial Phase
- Phase I
- Phase II
- Phase III
- Phase IV
By End User
- Pharmaceutical & Biotechnology Companies
- Contract Research Organizations
- Medical Device Manufacturers
- Academic & Research Institutions
- Other End Users
By Region
- North America
- Europe
- APAC
- Middle East and Africa
- LATAM
Frequently Asked Questions (FAQs)
Author's Details
Debashree B
Healthcare Lead
Debashree Bora is a Healthcare Lead with over 7 years of industry experience, specializing in Healthcare IT. She provides comprehensive market insights on digital health, electronic medical records, telehealth, and healthcare analytics. Debashree’s research supports organizations in adopting technology-driven healthcare solutions, improving patient care, and achieving operational efficiency in a rapidly transforming healthcare ecosystem.










