Self-testing Market Size, Share & Trends Analysis Report By Sample Type (Blood, Urine, Saliva, Nasal Swab, Others), By Application (Blood Pressure Tests, Diabetes and Glucose Tests, Cholesterol and Triglycerides Tests, Pregnancy Tests, Infectious Disease Tests, Urinary Tract Infection Tests, Anemia Tests, Others), By Distribution Channel (Retail Pharmacies & Drug Stores, Online Pharmacies & DTC Websites, Supermarkets/Hypermarkets, Others) and By Region (North America, Europe, APAC, Middle East and Africa, LATAM) Forecasts, 2026-2034
Self-testing Market Size
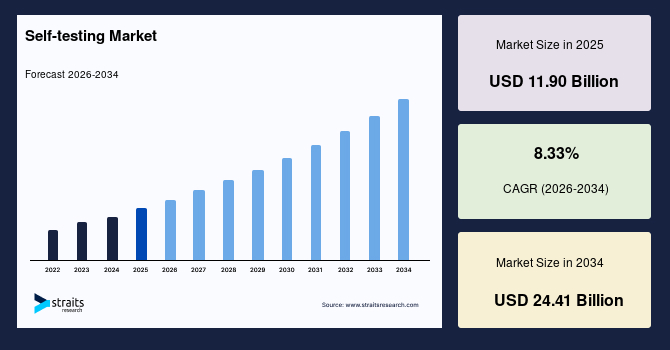
The self-testing market size was valued at USD 11.90 billion in 2025 and is projected to grow from USD 12.87 billion in 2026 to USD 24.41 billion by 2034 at a CAGR of 8.33% during the forecast period (2026-2034).
The self-testing market is growing rapidly as consumers increasingly prefer convenient, at-home diagnostic solutions for conditions such as diabetes, infectious diseases, pregnancy, fertility, cholesterol, and infectious disease screening. Rising awareness about early disease detection and preventive healthcare is driving strong demand, especially after the widespread adoption of COVID-19 self-test kits. In 2025, companies such as Abbott, Roche, and QuidelOrtho expanded their portfolios with advanced rapid diagnostic kits integrated with smartphone apps for real-time result tracking. Technological advancements in lateral flow assays, biosensors, and AI-powered interpretation tools are improving test accuracy and user confidence. Growth is further supported by the expansion of e-commerce pharmacies and telehealth platforms, which make self-testing products more accessible. However, challenges remain, including concerns about test accuracy for complex diseases, regulatory restrictions in some regions, and affordability issues in low-income markets, which may limit widespread adoption despite strong long-term growth potential.
Key Market Insights
- North America dominated the market with the largest share of 37.54% in 2025.
- The Asia Pacific is expected to be the fastest-growing region in the market during the forecast period, registering a CAGR of 10.15%.
- Based on sample type, the urine segment is expected to register a CAGR of 8.78% during the forecast period.
- Based on application, the infectious disease tests segment is projected to grow at a CAGR of 9.26% during the forecast period
- Based on distribution channel, the retail pharmacies & drug stores segment dominated the market, accounting for 40.82% revenue share in 2025.
- The US self-testing market size was valued at USD 4.02 billion in 2025 and is projected to reach USD 4.35 billion in 2026.
Market Summary
| Market Metric | Details & Data (2025-2034) |
|---|---|
| 2025 Market Valuation | USD 11.90 Billion |
| Estimated 2026 Value | USD 12.87 Billion |
| Projected 2034 Value | USD 24.41 Billion |
| CAGR (2026-2034) | 8.33% |
| Study Period | 2022-2034 |
| Dominant Region | North America |
| Fastest Growing Region | Asia Pacific |
| Key Market Players | Abbott, Hoffmann-La Roche AG, Siemens Healthineers, BD, Johnson & Johnson |
Download Free Sample Report to Get Detailed Insights.
Emerging Trends in Self-testing Market
Adoption of Smartphone-integrated Lateral Flow Reader Ecosystems
Self-testing is increasingly shifting toward smartphone-integrated diagnostic systems that convert simple test strips into digital health tools. Users scan lateral flow tests through mobile apps, which use AI-based image analysis and cloud processing to deliver more accurate and quantitative results. This reduces human reading errors and improves reliability for conditions like infectious diseases and hormone testing. For example, the Mobile Assay mReader platform turns a smartphone into a quantitative diagnostic reader for lateral flow test strips. This trend strengthens telehealth adoption, improves patient convenience, and transforms self-testing into a more data-driven and connected healthcare approach.
Adoption of CRISPR-based Ultra-sensitive Home Molecular Diagnostics
CRISPR-based self-testing platforms that enable molecular-level detection of pathogens and biomarkers at home without lab amplification steps boos market growth. Technologies such as SHERLOCK and DETECTR systems use gene-editing enzymes to identify viral RNA or DNA with high specificity in saliva or nasal samples. Startups like Mammoth Biosciences are advancing compact CRISPR test kits for infectious diseases and genetic markers. This innovation significantly improves early detection sensitivity compared to traditional antigen tests, enabling rapid, low-cost, and highly accurate self-diagnostics outside clinical environments.
Market Drivers
Expansion of At-home Molecular Sample Stabilization Kits and Rising Prevalence of Chronic and Infectious Diseases Drive Market
A key market driver is the growing use of at-home molecular sample stabilization kits that preserve sample quality for lab-based analysis after home collection. These systems use chemical preservatives or dried blood spot cards to stabilize saliva, blood, or swab samples during shipping without degradation. For example, the LetsGetChecked at-home STI testing kit, where users collect samples at home and send them to certified labs using stabilization media that maintains DNA/RNA integrity. This approach improves diagnostic accuracy, enables lab-grade testing remotely, and removes the need for cold-chain transport or immediate clinical handling.
The increasing global burden of chronic and infectious diseases is significantly raising demand for frequent and convenient home-based diagnostics. Conditions such as diabetes, hypertension, and sexually transmitted infections are becoming more widespread, pushing individuals toward regular self-monitoring solutions. For example, over 1 million STIs are acquired daily worldwide, highlighting the urgent need for accessible testing outside clinical settings. Similarly, rising diabetes prevalence is accelerating the adoption of at-home glucose monitoring kits. This growing disease burden encourages early detection and continuous monitoring and reduces dependence on hospitals, strongly driving market expansion.
Market Restraints
Limited Clinical Validation and Data Privacy and Cybersecurity Risks Restrain Market Growth
The lack of extensive clinical validation for emerging multi-condition home test panels that screen for multiple biomarkers restrains market growth. While single-condition tests like glucose or pregnancy are well established, broader panels for diseases such as thyroid disorders, STIs, or cardiovascular risk often face limited peer-reviewed validation and inconsistent sensitivity across populations. This raises concerns among healthcare providers about reliability and false positives. Regulatory bodies also impose stricter approval requirements, slowing commercialization and limiting adoption of advanced multiplex self-testing solutions in routine clinical practice and insurance-covered diagnostic pathways.
A key market restraint is the increasing vulnerability of connected health platforms to large-scale healthcare data breaches, which raises concerns over privacy and limits user trust in app-based diagnostic ecosystems. In 2025, a major breach affected approximately 529,000 individuals through Oracle Health systems, where attackers accessed medical and financial data such as diagnoses, insurance details, and personal identifiers. Such incidents highlight risks in digital diagnostic infrastructure that also supports self-testing apps and cloud-linked home diagnostics. Growing exposure of sensitive health data increases regulatory pressure and slows consumer adoption of connected self-testing solutions.
Market Opportunities
Expansion of Direct-to-Consumer Digital Diagnostic Platforms and Adoption of Precision Micro-coating Manufacturing Offer Growth Opportunities Market Players
The rapid growth of direct-to-consumer digital diagnostic platforms presents a strong opportunity in the self-testing market by enabling wider access to at-home medical testing services. Platforms such as Everlywell and LetsGetChecked allow consumers to conveniently order test kits, collect samples at home, and receive clinically validated results through digital apps without visiting healthcare facilities. This model creates opportunities for improved preventive care adoption, faster diagnosis, and better patient engagement. Integration with telehealth services further enhances treatment pathways, while subscription-based testing models open new revenue streams and support continuous health monitoring across large consumer populations.
A key opportunity for manufacturers in the self-testing market is the adoption of precision micro-coating manufacturing lines for large-scale production of lateral flow diagnostic strips. These advanced systems enable highly accurate and uniform deposition of biological reagents such as antibodies and enzymes onto nitrocellulose membranes, which is critical for ensuring consistent test performance. By using robotic dispensing, automated alignment, and high-resolution coating technologies, manufacturers can significantly improve product reliability and reduce batch-to-batch variation. This enhances test sensitivity and accuracy while lowering reagent wastage and production costs. It also helps manufacturers meet strict regulatory standards and scale efficiently for global demand.
Regional Insights
North America: Market Leadership Driven by Strong Penetration of Pharmacy-based Purchase and Telehealth Integration
The North America self-testing market accounted for a revenue share of 37.54% in 2025 due to strong penetration of pharmacy-based self-testing that directly guides consumers toward home diagnostic kits after initial screening. Retail chains like Walgreens in the US increasingly integrate in-store digital health stations that recommend follow-up at-home tests for cholesterol, flu, and STI screening. High adoption of employer-sponsored wellness programs, where companies such as Walmart and Amazon provide employees with subsidized at-home metabolic and infectious disease testing kits. Strong FDA Emergency Use Authorization (EUA) frameworks accelerate rapid commercialization of innovative self-testing products across the region.
The US market is growing due to an established diagnostic infrastructure, where users collect samples at home and send them to CLIA-certified labs using prepaid logistics, widely used by companies like Everlywell and LetsGetChecked. Rising use of HSA and FSA tax-advantaged spending accounts encourages consumers to purchase self-testing kits, improving affordability and accelerating routine preventive health testing adoption across the US healthcare system.
Canada's market benefits from provincial telehealth integration, where platforms like Ontario Health and Maple support remote consultation and recommend at-home diagnostic kits for conditions such as diabetes and infections. Strong public funding for preventive screening programs, where provinces distribute home-based kits for colorectal cancer screening, such as the FIT test program in Ontario and British Columbia. Rising demand for Indigenous and remote community healthcare access programs supports mail-in diagnostics and mobile health kits, improving early disease detection in underserved northern regions.
Asia Pacific: Fastest Growth Driven by High Prevalence of Infectious Diseases and Strong Penetration of Digital Health
The Asia Pacific self-testing market is expected to register the fastest growth with a CAGR of 10.15% during the forecast period. This growth is supported by a high prevalence of infectious and chronic diseases such as diabetes, tuberculosis, and hepatitis, especially in India and China, where patient pools are large and under-screened. For example, India alone has over 100 million diabetes patients, increasing demand for home glucose and metabolic self-tests. Strong local manufacturing capacity in China, India, and South Korea, where companies like Wondfo Biotech and SD Biosensor produce low-cost rapid test kits at scale, improving affordability and supply chain access across rural and urban Asia Pacific regions.
The Chinese market is growing due to strong government-led “Internet + Healthcare” policy integration, where platforms like Ping A Good Doctor and WeDoctor enable home diagnostic ordering and digital result interpretation. Large-scale domestic manufacturing of low-cost rapid test kits by companies, ensuring high supply availability. The widespread use of community health stations in tier-2 and tier-3 cities supports the distribution of infectious disease self-test kits, improving early screening and rural healthcare access across China.
The India self-testing market grows due to the strong penetration of digital health platforms such as Tata 1mg, PharmEasy, and Practo, which enable ordering and interpretation of at-home diagnostic kits across urban and semi-urban areas. Another key driver is expanding pharmacy retail networks like Apollo Pharmacy and MedPlus, which improve access to low-cost rapid test kits for diabetes, pregnancy, and infections. Government-backed initiatives such as the Ayushman Bharat Digital Mission support digital health records integration, encouraging adoption of home-based testing and telehealth-linked diagnostics across India.
By Sample Type
The blood segment dominated the market with a revenue share of 41.17% in 2025 due to high diagnostic reliability of capillary blood in glucose, cholesterol, and HbA1c monitoring. Finger-prick sampling enables quantitative biomarker detection suitable for chronic disease management. Wide adoption of glucose monitoring devices like Abbott FreeStyle Libre supports this segment. Improved micro-lancet and painless sampling technologies increase user compliance, making blood-based tests the preferred format for accurate home diagnostics.
The urine segment is expected to grow at a CAGR of 8.78% during the forecast period due to the strong suitability of urine-based biomarkers for non-invasive self-testing, especially in pregnancy, UTI, and fertility diagnostics. High stability of analytes like hCG enables accurate home pregnancy testing without blood sampling. Another factor is the improved sensitivity of modern lateral flow urine assays, reducing false negatives in infection screening. Increasing adoption of UTI self-test kits in pharmacies and e-commerce platforms supports frequent use.
By Application
The diabetes and glucose tests segment dominated the market, accounting for 29.05% revenue share in 2025 due to post-meal glucose self-testing adoption, where patients increasingly test blood sugar 1–2 hours after meals to manage diet-triggered spikes more precisely in daily life. Increasing cases of type 2 diabetes linked to sedentary lifestyles, obesity, and aging populations significantly expand test usage frequency, which, in turn, support market growth.
The infectious disease tests segment is expected to grow at a CAGR of 9.26% during the forecast period, driven by rising incidence of respiratory and vector-borne infections requiring rapid home screening, especially during seasonal flu peaks and post-pandemic surveillance. Expanding pharmacy and e-commerce distribution of STI self-test kits supports private, quick diagnosis, improving early detection and containment of infectious diseases outside clinical settings.
By Distribution Channel
The retail pharmacies & drug stores segment dominated the market, accounting for 40.82% revenue share in 2025. This dominance can be attributed to instant product availability and a zero-waiting-time purchase model, allowing consumers to buy pregnancy, glucose, and infectious disease kits immediately without online delivery delays. Strong integration of over-the-counter (OTC) regulatory approvals in pharmacies enables easy shelf placement of self-test kits, making pharmacies the primary distribution channel for rapid diagnostic products.
The online pharmacies & DTC websites segment is projected to grow at a CAGR of 9.83% during the forecast period, fueled by rising adoption of home-delivered diagnostic kits with cold-chain-free logistics, enabling seamless access to tests for hormones, STIs, and metabolic conditions without visiting physical stores. Digital prescription-free ordering for OTC self-test kits, where platforms like PharmEasy and Tata 1mg allow users to directly purchase and schedule repeat testing through apps.
Competitive Landscape
The self-testing market is fragmented with strong competition between global diagnostics companies and digital health players. Major companies such as Abbott, Roche, Siemens Healthineers, and QuidelOrtho dominate due to their wide range of rapid diagnostic kits, strong R&D capabilities, and global distribution networks. Consumer-focused companies like Everlywell, LetsGetChecked, and OraSure Technologies focus on direct-to-consumer testing models through online platforms. Emerging startups develop smartphone-integrated and AI-based self-testing solutions. Competition is driven by product accuracy, ease of use, digital connectivity, regulatory approvals, and expansion into chronic disease and infectious disease home testing segments across global markets.
List of Key and Emerging Players in Self-testing Market
- Abbott
- Hoffmann-La Roche AG
- Siemens Healthineers
- BD
- Johnson & Johnson
- QuidelOrtho Corporation
- Thermo Fisher Scientific
- Bio-Rad Laboratories
- Danaher Corporation
- Sysmex Corporation
- Qiagen N.V.
- bioMérieux
- OraSure Technologies
- Cardinal Health
- Geratherm Medical AG
- ACON Laboratories Inc.
- ARKRAY Inc.
- Insight Molecular Diagnostics Inc.
- Bionime Corporation
- Trinity Biotech plc
- OraSure
Recent Developments
- In March 2026, Insight Molecular Diagnostics Inc. submitted GraftAssureDx (a kitted molecular assay enabling decentralized testing) to the FDA for review, marking a shift toward near-patient/self-testing transplant monitoring.
- In January 2026, OraSure submitted FDA applications for an OTC rapid molecular self-test for Chlamydia and Gonorrhea (~30-minute results) along with the Colli-Pee at-home urine collection device, advancing decentralized STI diagnostics.
Report Scope
| Report Metric | Details |
|---|---|
| Market Size in 2025 | USD 11.90 Billion |
| Market Size in 2026 | USD 12.87 Billion |
| Market Size in 2034 | USD 24.41 Billion |
| CAGR | 8.33% (2026-2034) |
| Base Year for Estimation | 2025 |
| Historical Data | 2022-2024 |
| Forecast Period | 2026-2034 |
| Report Coverage | Revenue Forecast, Competitive Landscape, Growth Factors, Environment & Regulatory Landscape and Trends |
| Segments Covered | By Sample Type, By Application, By Distribution Channel |
| Geographies Covered | North America, Europe, APAC, Middle East and Africa, LATAM |
| Countries Covered | US, Canada, UK, Germany, France, Spain, Italy, Russia, Nordic, Benelux, China, Korea, Japan, India, Australia, Taiwan, South East Asia, UAE, Turkey, Saudi Arabia, South Africa, Egypt, Nigeria, Brazil, Mexico, Argentina, Chile, Colombia |
Download Free Sample Report to Get Detailed Insights.
Self-testing Market Segments
By Sample Type
- Blood
- Urine
- Saliva
- Nasal Swab
- Others
By Application
- Blood Pressure Tests
- Diabetes and Glucose Tests
- Cholesterol and Triglycerides Tests
- Pregnancy Tests
- Infectious Disease Tests
- Urinary Tract Infection Tests
- Anemia Tests
- Others
By Distribution Channel
- Retail Pharmacies & Drug Stores
- Online Pharmacies & DTC Websites
- Supermarkets/Hypermarkets
- Others
By Region
- North America
- Europe
- APAC
- Middle East and Africa
- LATAM
Frequently Asked Questions (FAQs)
Author's Details
Debashree B
Healthcare Lead
Debashree Bora is a Healthcare Lead with over 7 years of industry experience, specializing in Healthcare IT. She provides comprehensive market insights on digital health, electronic medical records, telehealth, and healthcare analytics. Debashree’s research supports organizations in adopting technology-driven healthcare solutions, improving patient care, and achieving operational efficiency in a rapidly transforming healthcare ecosystem.










