IVD Raw Materials Market Size, Share & Trends Analysis Report By Product (Antibody, Antigens, Proteins, Enzymes, Biological Buffers, Substrates, Others), By Technology (Molecular Diagnostics, Clinical Chemistry, Immunochemistry, Others), By End User (Diagnostic Laboratories, Pharmaceutical & Biotechnological Companies, Others) and By Region (North America, Europe, APAC, Middle East and Africa, LATAM) Forecasts, 2026-2034
What is the Size of IVD Raw Materials Market?
The IVD raw materials market size was valued at USD 18.09 billion in 2025 and is projected to reach USD 20.11 billion in 2026 to USD 48.27 billion by 2034, growing at a CAGR of 11.55% during the forecast period (2026-2034),.
Key Market Insights
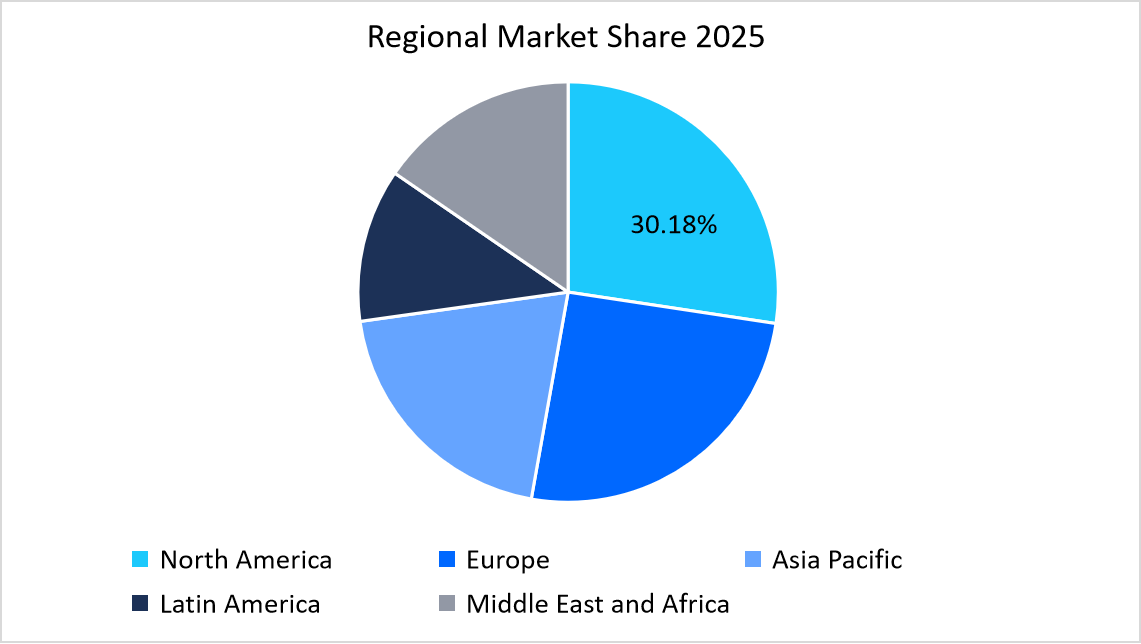
- North America dominated the IVD raw materials market with the largest share of 30.18% in 2025.
- The Asia Pacific region is expected to be the fastest-growing region in the IVD raw materials market during the forecast period, growing at a CAGR of 13.55%.
- By product, the antibody segment dominated the market in 2025 with a revenue share of 47.23%.
- By technology, the immunochemistry segment dominated the market with a revenue share of 34.54% in 2025.
- By end user, the pharmaceutical & biotechnology companies segment dominated the market with a revenue share of 40.33% in 2025.
- The US IVD raw materials market size was valued at USD 4.71 billion in 2025 and is projected to reach USD 5.23 billion in 2026.
Market Summary
| Market Metric | Details & Data (2025-2034) |
|---|---|
| 2025 Market Valuation | USD 18.09 Billion |
| Estimated 2026 Value | USD 20.11 Billion |
| Projected 2034 Value | USD 48.27 Billion |
| CAGR (2026-2034) | 11.55% |
| Study Period | 2022-2034 |
| Dominant Region | North America |
| Fastest Growing Region | Asia Pacific |
| Key Market Players | Creative Diagnostics, Merck KGaA, Thermo Fisher Scientific Inc, OYC Americas, Inc., Fapon Biotech Inc. |

Download Free Sample Report to Get Detailed Insights.
What are the Current Trends in IVD Raw Materials Market?
IVD manufacturers are increasingly demanding highly purified antibodies, recombinant proteins, enzymes, and antigens to improve assay sensitivity, specificity, and reproducibility. With rising regulatory protocols, raw material suppliers are investing in stringent quality control, traceability systems, and GMP-compliant production processes to improve diagnostic reliability.
The continued expansion of PCR, real-time PCR, digital PCR, and next-generation sequencing-based diagnostics is accelerating demand for thermostable polymerases, reverse transcriptase, nucleotides, and molecular-grade buffers. Organizations continue to promote molecular testing for infectious diseases and antimicrobial resistance surveillance, reinforcing the need for high-performance nucleic acid amplification components.
A shift from animal-sourced antibodies and proteins toward recombinant monoclonal antibodies and synthetic peptides to ensure batch-to-batch consistency and reduce biological variability presents a major market trend. Regulatory authorities emphasize consistency and risk mitigation in diagnostic manufacturing, encouraging the use of well-characterized recombinant materials.
IVD manufacturers are designing automated high-throughput platforms for clinical laboratories, which increases demand for raw materials that are stable, preformulated, and compatible with closed-system analyzers. Companies are developing lyophilized beads, stabilized liquid reagents, and pre-conjugated antibodies that reduce in-house processing requirements in order to reduce manufacturing complexity for diagnostic kits.
What are the Key Drivers of IVD Raw Materials Market?
The increasing prevalence of chronic diseases and infectious diseases is driving higher diagnostic testing volumes worldwide. According to the WHO, noncommunicable diseases account for the majority of global deaths, reinforcing the need for early detection and continuous monitoring. Higher test volumes directly increase demand for antibodies, antigens, enzymes, membranes, and assay substrates used in diagnostic kits. This sustained clinical demand expands consumption of IVD raw materials across hospitals, laboratories, and public health programs.
Governments and healthcare systems are investing in preventive diagnostics and population-level screening initiatives for cervical cancer, hepatitis, HIV, and metabolic disorders. As screening programs expand to rural and semi-urban regions, demand for large-scale production of diagnostic kits rises accordingly. This growth directly stimulates procurement of bulk raw materials, including immunoassay reagents, buffers, and substrates.
Point-of-care diagnostics for glucose monitoring, cardiac markers, infectious diseases, and pregnancy testing are expanding rapidly in both developed and emerging economies. This expansion increases the manufacturing output of rapid diagnostic kits and lateral flow assays, which rely on membranes, conjugates, labels, and specialty chemicals. As access to diagnostics improves at the primary care level, raw material consumption rises proportionally, which propels market growth.
What are the Restraining Factors in IVD Raw Materials Market?
Polyclonal antibodies, native antigens, and enzyme preparations remain sensitive to source conditions, cell lines, animal hosts, and purification methods, which lead to batch-to-batch performance variation. This variability forces IVD manufacturers to conduct additional quality checks and bridging studies, increasing costs, slowing production cycles, and reducing manufacturing efficiency.
Proprietary technologies and licensing restrictions limit sourcing options and prevent alternative material development by new or smaller players. This reduces competitive pricing, slows innovation timelines, restricts supplier diversification, and lowers overall market openness, which negatively affects adoption and long-term market growth.
Antibody and biological reagent production depends on complex upstream inputs such as cell culture media, purification resins, and rare biochemical substrates, which face price fluctuations and capacity constraints. This volatility creates pricing instability and procurement uncertainty, weakens margin predictability, discourages long-term supply contracts, and slows strategic investments, thereby restraining market expansion.
What are the Emerging Opportunities in IVD Raw Materials Market?
Growth of companion diagnostics and precision medicine creates demand for highly characterized biomarker-specific antibodies, recombinant proteins, and calibrators, which presents as a market opportunity. The pharmaceutical companies expand precision medicine pipelines through which raw material suppliers can establish strategic partnerships for co-development projects, which hence opens new avenues for IVD raw material manufacturers.
Expansion of veterinary and animal health diagnostics accelerates demand for veterinary diagnostic testing, increasing production of veterinary immunoassays and molecular tests. This requires species-specific antigens, antibodies, and reagents and provides room for product differentiation and portfolio expansion, which presents a scalable opportunity for IVD raw material suppliers.
Development of next-generation biosensor and microfluidic platforms creates demand for novel surface chemistries, nanomaterials, conductive inks, and functionalized biomolecules. Research institutions and MedTech innovators are increasingly integrating biochemical reagents with miniaturized detection systems for rapid diagnostics. These platforms require specialized raw materials with enhanced binding efficiency and stability under low-volume conditions. Suppliers that invest in material science capabilities capture early-stage design for next-generation diagnostic devices, which opens new application-driven revenue streams.
Regional Analysis
North America IVD Raw Materials Market
The IVD raw materials market in North America held the largest share of 30.18% in 2025 due to widespread use of molecular diagnostics immunoassays and clinical chemistry testing across hospital laboratories, reference laboratories and integrated diagnostic networks. Diagnostic manufacturers in the region emphasize stable access to enzymes, antibodies buffers and biochemical reagents to support continuous production of automated assay kits. Regulatory alignment with quality system expectations further reinforces long-term sourcing relationships between raw material suppliers and diagnostic kit developers.
Asia Pacific IVD Raw Materials Market
The IVD raw materials market in Asia Pacific is expected to grow at a CAGR of 13.55% during the forecast period. This growth is attributed to rising diagnostic testing volumes, public health laboratory upgrades, and government-supported localization of assay manufacturing. Countries across the region promote domestic production of enzymes primers probes and immunoreagents to reduce reliance on imports and strengthen diagnostic self-sufficiency. Growth in centralized laboratories and automated testing platforms further increases consumption of consistent raw material inputs.
Source: Straits Analysis
Europe IVD Raw Materials Market
The Europe market is shaped by structured diagnostic pathways and increasing alignment with region-wide regulatory requirements governing in vitro diagnostics. Diagnostic developers across the region prioritize traceable sourcing of biological and chemical inputs to support conformity assessment and technical documentation. Public laboratories and academic medical centers contribute to sustained consumption of antibodies enzymes and reference materials across oncology autoimmune and infectious disease testing, which further enhances the market growth.
Middle East & Africa IVD Raw Materials Market
The Middle East & Africa market evolves through expansion of public sector laboratories and national diagnostic capacity building initiatives. Government-funded health systems prioritize dependable access to assay reagents and consumables for infectious disease surveillance blood screening and chronic disease monitoring. Centralized procurement frameworks influence sourcing volumes for antibodies enzymes and biochemical components across public laboratories, which enhances market growth.
Latin America IVD Raw Materials Market
The growth in Latin America is supported by public healthcare investment focused on laboratory diagnostics for chronic diseases maternal health and infectious disease control. National laboratory networks rely on centralized purchasing systems, which shape volume-driven sourcing of enzymes antibodies and clinical chemistry reagents. Deployment of automated analyzers across tertiary hospitals sustains recurring demand for standardized reagent inputs, which further fuels the market growth.
Product Insights
Antibodies dominated the product segment with a revenue share of 47.23%. The growth is attributed to its extensive use in immunoassays, rapid diagnostic tests, ELISA kits, and lateral flow assays, which further contribute to its largest market share.
The proteins segment is projected to grow at a CAGR of 12.44% during the forecast period. The rising application of recombinant proteins as biomarkers, controls, and calibrators in advanced diagnostic platforms is fueling segment growth. Expanding research activities and increasing development of novel diagnostic assays further support the rapid expansion of the proteins segment.
Technology Insights
Immunochemistry held the largest market share of 34.54% in 2025 due to the widespread adoption of immunoassay-based diagnostic techniques for detecting hormones, infectious agents, cardiac markers, and tumor markers.
Molecular diagnostics is anticipated to grow at a CAGR of 12.67% during the forecast period. Increasing demand for early disease detection, personalized medicine, and genetic testing is driving the adoption of PCR, nucleic acid amplification tests, and next-generation sequencing technologies. A growing focus on precision diagnostics further accelerates the growth of the molecular diagnostics segment.
End User Insights
Pharmaceutical and biotechnological companies led the end-user segment with a share of 40.33% in 2025 because of the extensive utilization of IVD raw materials in drug discovery, biomarker validation, companion diagnostics, and clinical trials.
The diagnostic laboratories segment is projected to grow at a CAGR of 12.85% during the forecast period due to the rising volume of diagnostic testing, increasing automation in laboratories, and expanding demand for accurate and rapid tests. The growing burden of chronic and infectious diseases further contributes to the strong growth of this segment.
Competitive Landscape
The global IVD raw materials market is moderately concentrated, characterized by the presence of established biochemical and diagnostics suppliers alongside specialized niche manufacturers focused on enzymes, antibodies, antigens, and molecular reagents. Market competition is framed by parameters such as material performance consistency, compatibility with immunoassay molecules, and suitability for automated workflows. Leading players maintain their market position through broad raw material portfolios, spanning enzymes substrates buffers antibodies and nucleic acid components, supported by strong quality assurance systems, regulatory compliance capabilities, and long-term supply partnerships with diagnostic kit manufacturers and OEMs.
List of Key and Emerging Players in IVD Raw Materials Market
- Creative Diagnostics
- Merck KGaA
- Thermo Fisher Scientific Inc
- OYC Americas, Inc.
- Fapon Biotech Inc.
- Biosynth
- Hoffmann-La Roche Ltd
- Sysmex Corporation
- Shenzhen AIVD Biotechnology Co., Ltd.
- Glentham Life Sciences Limited
- Neogen Corporation
- MP Biomedicals
- Angstrom Biotech
- SR Bioera
- Bio-Rad Laboratories
- Fujirebio Holdings, Inc.
- Sysmex Corporation
Latest News on Key Players and Emerging Players
| TIMELINE | COMPANY | DEVELOPMENT |
|---|---|---|
|
February 2026 |
Fujirebio Holdings, Inc. |
Fujirebio Holdings, Inc. and Sysmex Corporation announced a strategic sales collaboration focused on dementia diagnostic testing solutions. Under the agreement, Sysmex expands global distribution of Fujirebio’s Lumipulse immunoassay systems and related reagents in selected regions. |
|
June 2025 |
Fujirebio Holdings, Inc. |
Fujirebio acquired Plasma Services Group to strengthen its position as a provider of critical and high-quality biological raw materials to the IVD and life science industries. |
Source: Secondary Research
Report Scope
| Report Metric | Details |
|---|---|
| Market Size in 2025 | USD 18.09 Billion |
| Market Size in 2026 | USD 20.11 Billion |
| Market Size in 2034 | USD 48.27 Billion |
| CAGR | 11.55% (2026-2034) |
| Base Year for Estimation | 2025 |
| Historical Data | 2022-2024 |
| Forecast Period | 2026-2034 |
| Report Coverage | Revenue Forecast, Competitive Landscape, Growth Factors, Environment & Regulatory Landscape and Trends |
| Segments Covered | By Product, By Technology, By End User |
| Geographies Covered | North America, Europe, APAC, Middle East and Africa, LATAM |
| Countries Covered | US, Canada, UK, Germany, France, Spain, Italy, Russia, Nordic, Benelux, China, Korea, Japan, India, Australia, Singapore, Taiwan, South East Asia, UAE, Turkey, Saudi Arabia, South Africa, Egypt, Nigeria, Brazil, Mexico, Argentina, Chile, Colombia |
Download Free Sample Report to Get Detailed Insights.
IVD Raw Materials Market Segments
By Product
- Antibody
- Antigens
- Proteins
- Enzymes
- Biological Buffers
- Substrates
- Others
By Technology
- Molecular Diagnostics
- Clinical Chemistry
- Immunochemistry
- Others
By End User
- Diagnostic Laboratories
- Pharmaceutical & Biotechnological Companies
- Others
By Region
- North America
- Europe
- APAC
- Middle East and Africa
- LATAM
Frequently Asked Questions (FAQs)
Author's Details
Mitiksha Koul
Research Associate
Mitiksha Koul is a Research Associate with 2 years of experience in market research. She focuses on analyzing industry trends, competitive landscapes, and growth opportunities to support strategic decision-making. Mitiksha’s strong analytical skills and research expertise enable her to deliver actionable insights that help businesses adapt to evolving market dynamics and achieve sustainable growth.










